BASIC STOICHIOMETRY T01/s11 Based on the balanced equation, Molar Mass (g/mol) 21205 + 10NO 5N204 + 212 I205 333.80 calculate the molecules of NO required when 45 molecules of N,04 are formed. NO 30.006 N204 I2 Avogadro's No. 6.022x1023 mol"1 92.011 253.8 exact number, no tolerance Question Attempts: 0 of 1 used SUBMIT ANSWER SAVE FOR LATER BASIC STOICHIOMETRY TO1/s11 Based on the balanced equation, Molar Mass (g/mol) 2C2H6 + 702 - 4CO2 + 6H20 C2H6 02 CO2 H20 Avogadro's No. 6.022x1023 molr1 30.07 calculate the molecules of H20 formed when 119 molecules of 02 are reacted. 32.000 44.010 18.015 exact number, no tolerance Question Attempts: 0 of 1 used SUBMIT ANSWER SAVE FOR LATER
BASIC STOICHIOMETRY T01/s11 Based on the balanced equation, Molar Mass (g/mol) 21205 + 10NO 5N204 + 212 I205 333.80 calculate the molecules of NO required when 45 molecules of N,04 are formed. NO 30.006 N204 I2 Avogadro's No. 6.022x1023 mol"1 92.011 253.8 exact number, no tolerance Question Attempts: 0 of 1 used SUBMIT ANSWER SAVE FOR LATER BASIC STOICHIOMETRY TO1/s11 Based on the balanced equation, Molar Mass (g/mol) 2C2H6 + 702 - 4CO2 + 6H20 C2H6 02 CO2 H20 Avogadro's No. 6.022x1023 molr1 30.07 calculate the molecules of H20 formed when 119 molecules of 02 are reacted. 32.000 44.010 18.015 exact number, no tolerance Question Attempts: 0 of 1 used SUBMIT ANSWER SAVE FOR LATER
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter9: Chemical Quantities
Section: Chapter Questions
Problem 81AP: For each of the following incomplete and unbalanced equations, indicate how many moles of the second...
Related questions
Question
can you solve these 2 and just provide the answer I solved most of the work I just wanna make sure to get the answer right before i submit it

Transcribed Image Text:BASIC STOICHIOMETRY T01/s11
Based on the balanced equation,
Molar Mass (g/mol)
21205 + 10NO
5N204 + 212
I205
333.80
calculate the molecules of NO required when 45 molecules of N,04 are formed.
NO
30.006
N204
I2
Avogadro's No.
6.022x1023 mol"1
92.011
253.8
exact number, no tolerance
Question Attempts: 0 of 1 used
SUBMIT ANSWER
SAVE FOR LATER
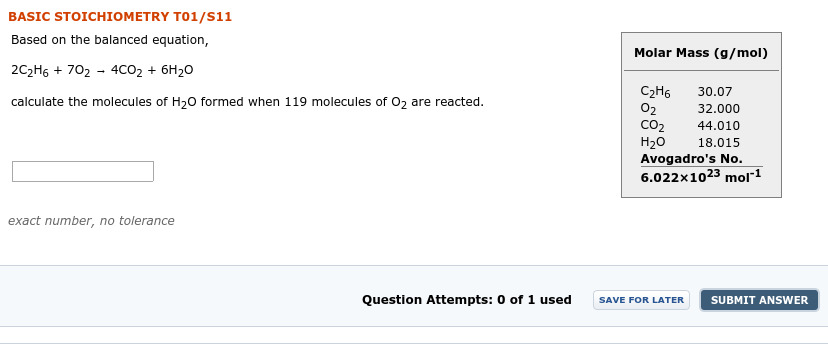
Transcribed Image Text:BASIC STOICHIOMETRY TO1/s11
Based on the balanced equation,
Molar Mass (g/mol)
2C2H6 + 702 - 4CO2 + 6H20
C2H6
02
CO2
H20
Avogadro's No.
6.022x1023 molr1
30.07
calculate the molecules of H20 formed when 119 molecules of 02 are reacted.
32.000
44.010
18.015
exact number, no tolerance
Question Attempts: 0 of 1 used
SUBMIT ANSWER
SAVE FOR LATER
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning