Boyle's Law states that when a sample of gas is compressed at a constant temperature, the pressure P and volume V satisfy the equation PV = C, where Cis a constant. Suppose that at a certain instant the volume is 100 cm³, the pressure is 100 kPa, and the pressure is increasing at a rate of 40 kPa/min. At what rate is the volume decreasing at this instant? |× cm³/min
Boyle's Law states that when a sample of gas is compressed at a constant temperature, the pressure P and volume V satisfy the equation PV = C, where Cis a constant. Suppose that at a certain instant the volume is 100 cm³, the pressure is 100 kPa, and the pressure is increasing at a rate of 40 kPa/min. At what rate is the volume decreasing at this instant? |× cm³/min
Linear Algebra: A Modern Introduction
4th Edition
ISBN:9781285463247
Author:David Poole
Publisher:David Poole
Chapter6: Vector Spaces
Section6.7: Applications
Problem 13EQ
Related questions
Question
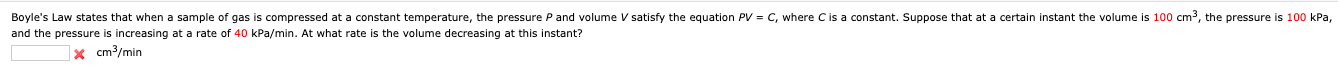
Transcribed Image Text:Boyle's Law states that when a sample of gas is compressed at a constant temperature, the pressure P and volume V satisfy the equation PV = C, where Cis a constant. Suppose that at a certain instant the volume is 100 cm³, the pressure is 100 kPa,
and the pressure is increasing at a rate of 40 kPa/min. At what rate is the volume decreasing at this instant?
|× cm³/min
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Linear Algebra: A Modern Introduction
Algebra
ISBN:
9781285463247
Author:
David Poole
Publisher:
Cengage Learning

Algebra & Trigonometry with Analytic Geometry
Algebra
ISBN:
9781133382119
Author:
Swokowski
Publisher:
Cengage

Linear Algebra: A Modern Introduction
Algebra
ISBN:
9781285463247
Author:
David Poole
Publisher:
Cengage Learning

Algebra & Trigonometry with Analytic Geometry
Algebra
ISBN:
9781133382119
Author:
Swokowski
Publisher:
Cengage