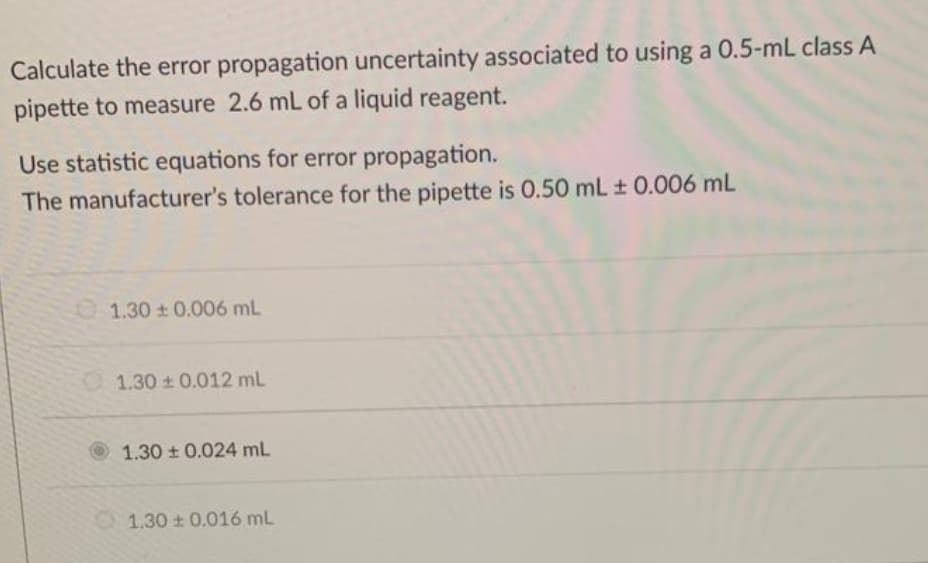
Calculate the error propagation uncertainty associated to using a 0.5-mL class A pipette to measure 2.6 mL of a liquid reagent. Use statistic equations for error propagation. The manufacturer's tolerance for the pipette is 0.50 mL ± 0.006 mL
Q: A method for the analysis of Ca2+ in water suffers from an interference in the presence of Zn2+.…
A: Consider the concentration of Zn+2 = 1 The concentration of Ca+2 = 100 The given…
Q: Describe an electrochemical sensor assay method for rapid bacterial detection and identification.…
A:
Q: A 300 g of a sample is analyzed by series of sieving. The results report were aperture (micrometer)…
A: In chemistry, there are many ways to check the deviation of experimental data from the accurate…
Q: Prepare standard solutions for the spectrophotometric measurement of iron in an unknown solution.…
A: Serial dilution refers to stepwise dilution by keeping the dilution factor constant. Dilution factor…
Q: 1. What's the slope of the calibration curve? 3.95 x 10-5 4.17x10-5 1 20484 25257 2. What's the…
A: The linear regression equation of the calibration curve is: Y = 25257X - 114.32
Q: Chloroform is an internal standard in the determination of the pesticide DDT in a polarographic…
A:
Q: Drinking water with negligible lead content was fortified with 2.28 µg Pb(II)/L. Triplicate…
A: Given: Concentration of drinking water with negligible Pb content = 2.28 µg Pb(II)/L Mean of…
Q: A 10.00-g sample containing an analyte was transferred to a 250-mL volumetric flask and į diluted to…
A: Solution - According to the question - Given - In the spiked sample, the signal is due to 10 ml of…
Q: The calibration curve for FD&C yellow #5 dye has a slope of 35188 L/mol with an intercept of 0.007.…
A: Given: Calibration curve for FD&C yellow #5 dye has Slope = 35188 L/mol Intercept = 0.007…
Q: The amount of caffeine in an analgesic tablet was determined by HPLC using a normal calibration…
A: A question based on quantitative analysis, which is to be accomplished.
Q: A potassium hydrogen phthalate solution was prepared by adding 0.0996 g of dry potassium hydrogen…
A: Here given that, Mass of dry potassium hydrogen phthalate = 0.0996 g So number of mols of…
Q: Quantitative Analysis by External Standard Method Calcium in a juice sample is determined by atomic…
A: Initial concentration of solution = M1 Volume of starting solution used = V1 Concentration of…
Q: Pure water containing no arsenic was spiked with 0.40 µg arsenate (AsO43) / L. Seven replicate…
A: The given information is represented as follows:
Q: A student wanted to determine the level of lead in amoxicillin powder, so he transferred 148 mg of…
A: Given: The absorbance of the diluted solution, y = 0.48 Calibration equation is given by y = 12.25…
Q: 2) You have just finished a GC experiment using the standard additions method to determine the…
A: Here some important data is missing hence i am doing it on general basics . "Since there are…
Q: You prepare several dilutions of an unknown compound. You measure the absorbance of each solution at…
A: The graph for concentration-Absorbance is shown below in which slope of the curve is determined.
Q: A solution of Ba (OH), was standardized against 0.1208 g of primary-standard-grade benzoic acid,…
A: Given 0.1208 g of benzoic acid, volume of base = 43.94g We have to find molar concentration of base…
Q: Using this table for the standard durve of cobalt (II) solution in UV Vis Molarity (M) Absorbance…
A: Given data L=10 cm we know that Beer Law is Absorbance=ε×M×Lwhere : M=Molarity , L=Length ,…
Q: A toluene solution (1.50 mg / L) shows an absorbance of 0.30 in a cuvette with a 0.025 mm optical…
A: The given data contains, Absorbance of toluene = 0.30 Pathlength of cuvette = 0.025 mm.…
Q: Please can you solve this question in chemistry quickly
A:
Q: The nitrate in water was analyzed using a spectrophotometric method. The absorbances of the…
A: Given data:
Q: The ratio of the number of atoms of the isotopes 69Ga and 71Ga in eight samples from different…
A:
Q: (b) Based on the performance and validation methods, explain the distinctions between the two terms…
A: Interpretation - As you already mentioned in the question that you want answer only question iii,…
Q: How would i perform a serial dilution of 1/2 and 1/4 if i have 7.5mL of an unknown protein, the…
A: The molarity of a solution is the number of moles of solute in the solvent. Molarity varies with the…
Q: Fluoride determination can be performed photometrically with alizarin red and Zirconium acid…
A: A question based on Beer-Lambert law, which is to be accomplished.
Q: II. Determination of Total Hardness of Water Source of water sample: Volume of water sample 22.00 mL…
A: Given that, Volume of EDTA solution consumed (V1)=14.06mL Molarity of standard EDTA solution used…
Q: In the Analytical Chemistry laboratory, one of the B.Sc student Ms. Fatma wanted to analyze the…
A: Spectrophotometer
Q: specific absorbance ratio
A: A1=€1cl and A2=€2cl A1/A2=€1/€2
Q: Blue Blue dye stock solution 0.293 M Absorbance at 630 nm 0.00265 Calibration curve y = 0.0833x A…
A: a) Calculation of theoretical molar concentration: MinitialVinitial=MfinalVfinal0.293 M×2.79…
Q: and unchlorinated drinking water. The results ± standa deviation (number of samples) are Sample…
A: Chromatography and spectrophotometry are the two methods used to measure nitrite content in rain…
Q: 2 1.2 1.1 3 0.9 4 0.7 For each set, determine the mean, spread, stand rariation. Calculate the…
A:
Q: The density of a solution was determined experimentally. The results of three calculations are:…
A: Density(D):- It is the ratio of mass of substance(in g) and the volume(in mL). Average density(Z):-…
Q: (b) Based on the performance and validation methods, explain the distinctions between the two tems…
A: ANSWER (b) (i) Linearity , is the property of a mathematical relationship (function) that can be…
Q: A set of samples from an abandoned industrial site detected a suspected groundwater contaminant at a…
A:
Q: Internal standard (As , Cs ), analyte with known concentration (Ax , Cx ) In a chromatographic…
A: Formula used: Area of analyte signalConc of analyte= F×Area of standard signalConc of…
Q: replicate determination of the lead content of a blood sample (0.752, 0.756, 0.752, 0.751 and 0.760…
A:
Q: A graphite furnace atomic absorption spectrophotometer (AAS) was used to determine the arsenic (As)…
A: Given data Concentration Absorbance 2 0.17 4 0.28 6 0.48 8 0.53 10 0.72
Q: Since this standard deviation value is close to the ϭ value, the iron concentration of 18.5 µg Fe/mL…
A:
Q: THE EXPERIMENTAL WRITE-UP AND COMPLETE THE PRE-LAB PRIOR TO DOW umber is: 620 n intensity of 4.06 n…
A: According to the question, we have a Quinine calibration curve and we neee to determine the.…
Q: . To determine the concentration of analyte in a sample a standard additions was performed. A…
A: Analyse the data and with given information, sketch the correct graph.
Q: The following results were obtained for the determination of calcium in a NIST limestone sample:…
A:
Q: In the Analytical Chemistry laboratory, one of the B.Sc student Ms. Fatma wanted to analyze the…
A: (i) Given: Dilution factor is 10. The volume of solution B(diluted solution) is 250 mL.…
Q: A student weighed out 0.150 g of protein powder and dissolved it in 100 mL of water (Solution 1).…
A: The percentage protein by mass in the original protein powder is to be calculated.
Q: (b) Based on the performance and validation methods, explain the distinctions between the two terms…
A:
Q: A student weighed out 0.150 g of protein powder and dissolved it in 100 mL of water (Solution 1).…
A:
Q: You are developing a procedure for determining traces of copper in biological materials using a wet…
A: To calculate the 95% confidence level for the above sample. Confidence interval = X ±ZσnX = mean…
Q: Descriptives Bronchial reactivity 95% Confidence Interval for Mean Lower Bound Upper Bound Minimum…
A:
Q: A standard curve for glucose analysis was prepared.The slope (m) was found to be 1.341 while the…
A: Given: Slope(m) = 1.341 y-intercept = -0.333 Absorbance (y) = 0.151
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- You are developing a procedure for determining traces of copper materials using a wet digestion followed by measurement by atomic absorption spectrophotometry. In order to test the validity of the method, you obtain an NIST orchard leaves standard reference material and analyze this material. Five replicas are sampled and analyzed, and the mean results are found to be 10.8 ppm with a standard deviation of plus or minus 0.7. ppm. The listed value for the standard reference is 11.7 ppm. Does your method give a statistically correct value at the 95% confidence level?You are developing a procedure for determining traces of copper in biological materials using a wet digestion followed by measurements by atomic absorption spectrophotometry. In order to test the validity of the method, you obtain a NIST orchard leaves standard reference material and analyze this material with your procedure five times and obtain a mean of 12.27 ppm with a standard deviation of 0.097 ppm. The NIST sample is listed as 11.7 ppm. Does your method give a statistically valid value at the 95% confidence level?Concentration=2.5 X 10^3 μM OD400nm=0.859 1cm cuvette is used what is the extinction coefficient (μM^-1 cm^-1)
- The following results were obtained for the determination of calcium in a NIST limestone sample: %CaO = 50.33, 50.22, 50.36, 50.21, and 50.44. Five gross samples were then obtained for a carload of limestone. The average %CaO values for the gross samples were found to be 49.53, 50.12, 49.60, 49.87, and 50.49. Calculate the relative standard deviation associated with the sampling stepExample: A GC-FID analysis was conducted on a soil sample containing pollutant X. The following separations were conducted: t (minutes) peak area Injection 1 21.1 ppm Toluene internal Standard 10.11 36,242 33.4 ppm 14.82 45,997 Injection 2 21.1 ppm Toluene Internal Standard 10.05 38,774 unknown concentration X 14.77 39,115 What is the concentration of X in the sample?1 mL was taken from the sample filtrate and mixed with 13 mL pure water and 4 mL sulfomolybdic acid and 2 mL dilute SnCI2 solution by adding it, and after waiting for 15 minutes, the absorbance of the resulting solutions against pure water was read at 520 nm. if the function of the calibration graph obtained with standard phosphorus solutions of 0.5-2.5 mg/mL is y= 0.245x + 0.107 and the absorbance of the serum sample is 0.342, how many grams of phosphorus is the amount in the sample?
- In quality management, it is very important that you have a good background knowledge in both descriptive and inferential statistics. Since you already took up Biostatistics and Epidemiology during your Second Year, answer BRIEFLY the following questions on basic statistics: 1. Compare and contrast the three most common measures of central tendency mean, median and mode. 2. Differentiate standard deviation from coefficient of variation. 3. What is T-test?The same chloride analysis but using a new method for which the standard deviation was not known, gave the following replicate results, mean and estimated standard deviation: Chloride (ppm) = 346, 359, 350; Mean = 351.67 ppm; Standard deviation = 6.66ppm. Solve for the CI for the 3 determinations at 95% probability level.You prepare the following MeOH standards from a 2000 ppm stock MeOH solution:Calibration Blank, 50 ppm, 100 ppm, 150 ppm and 400 ppm and analyze them using the GC-FID. You received the following data: Concentration of MeOH(ppm) Response (area) of MeOH peak (pA) 0 0 50 150 100 320 150 440 400 1100 Using the data above, make a properly documented graph of "Response (area)”versus "Concentration of MeOH (ppm)" and determine the linear workingrange (in units of concentration) of MeOH. This graph is to be done usingExcel. Be sure to provide a trendline, formula & R2 value).
- The internal standard method compensates for errors that affect both the analyte and the reference. True FalseA 0.0200 gram blood sample was decomposed by a microwave digestion technique followed by dilution to 100.0 mL in a volumetric flask. Aliquots of the sample solution were treated with a lead complexing reagent and water as follows: Solution 1: 10.0 ml blood sample + 20.0 mL complexing agent + 30.0 mL H20. Solution 2: 10.0 ml blood sample + 20.0 mL complexing agent + 26.0 mL H20 + 4.00 mL of 78 ppb Pb2+ standard. The resulting solutions were analyzed by UV/Vis at 375 nm. Absorbance for solution 1 = 0.155 and for solution 2 = 0.216. Calculate the concentration of lead (ppb) in the original sample.FT-IR technique can be utilized for the analysis of unknown analytes by matching it with__________. Both choices are correct Commercial library/database Reference standard



