Carbon dioxide and water react to form methanol and oxygen, like this: 2co,(9)+4H,0(9)→2 CH; OH()+30,(9) At a certain temperature, a chemist finds that a 6.5 L reaction vessel containing a mixture of carbon dioxide, water, methanol, and oxygen at equilibrium has the following composition: compound amount olo co2 3.58 g H,0 4.47 g Ar CH; OH 3.09 g 2.05 g Calculate the value of the equilibrium constant K, for this reaction. Round your answer to 2 significant digits. K = 0 %3D
Carbon dioxide and water react to form methanol and oxygen, like this: 2co,(9)+4H,0(9)→2 CH; OH()+30,(9) At a certain temperature, a chemist finds that a 6.5 L reaction vessel containing a mixture of carbon dioxide, water, methanol, and oxygen at equilibrium has the following composition: compound amount olo co2 3.58 g H,0 4.47 g Ar CH; OH 3.09 g 2.05 g Calculate the value of the equilibrium constant K, for this reaction. Round your answer to 2 significant digits. K = 0 %3D
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter19: The Chemistry Of The Main-group Elements
Section: Chapter Questions
Problem 60QRT
Related questions
Question
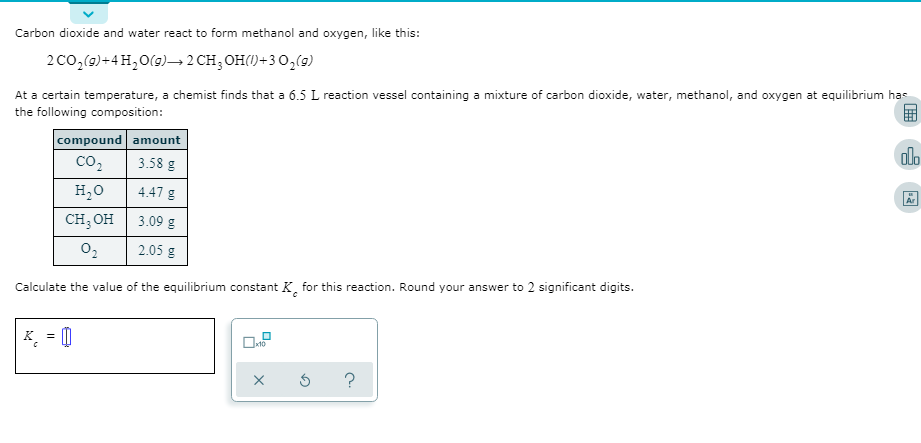
Transcribed Image Text:Carbon dioxide and water react to form methanol and oxygen, like this:
2co,(9)+4H,0(9)→2 CH; OH()+30,(9)
At a certain temperature, a chemist finds that a 6.5 L reaction vessel containing a mixture of carbon dioxide, water, methanol, and oxygen at equilibrium has
the following composition:
compound amount
olo
co2
3.58 g
H,0
4.47 g
Ar
CH; OH
3.09 g
2.05 g
Calculate the value of the equilibrium constant K, for this reaction. Round your answer to 2 significant digits.
K = 0
%3D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning