Chapter32: Gas Chromatography
Section: Chapter Questions
Problem 32.15QAP
Related questions
Question
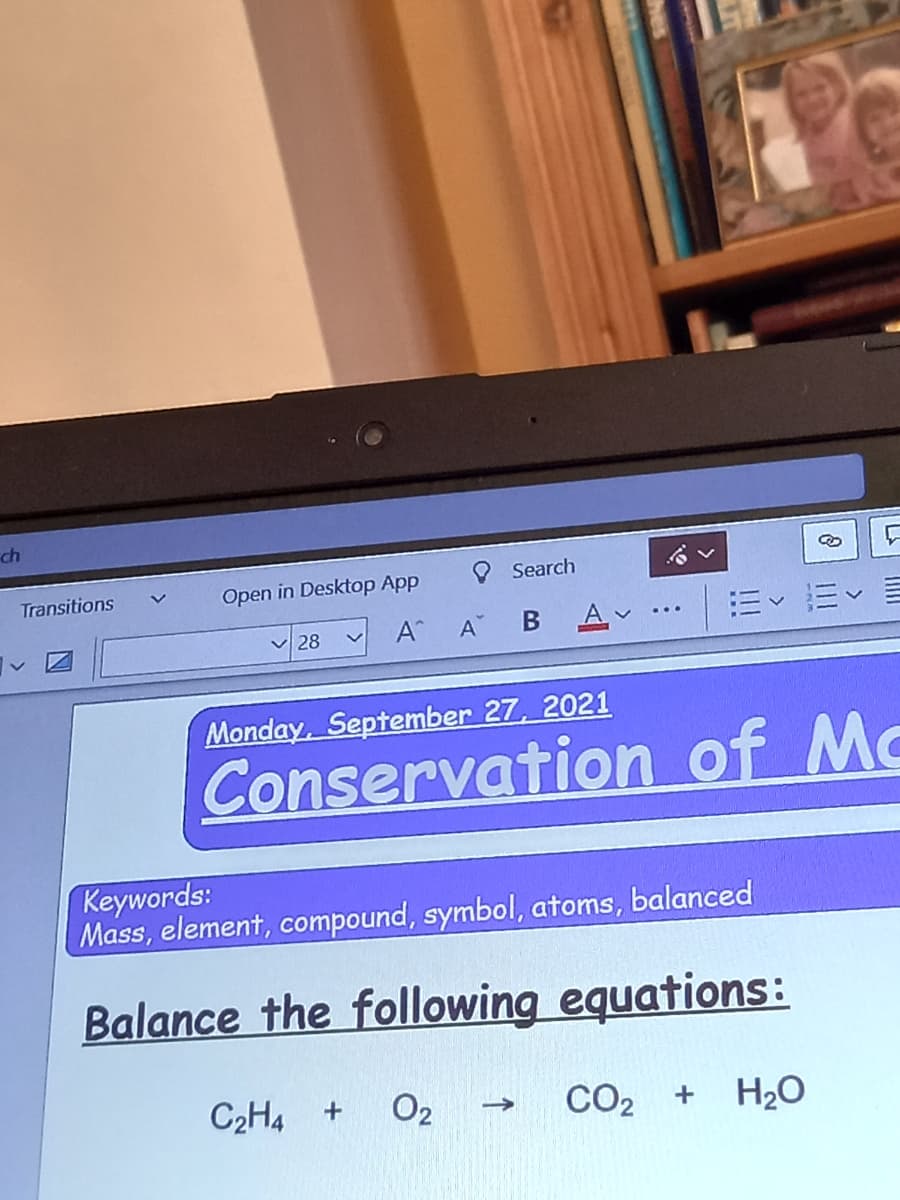
How do I balance it
Transcribed Image Text:ch
Search
Transitions
Open in Desktop App
A A B A ..
m<而<
V 28
Monday, September 27, 2021
Conservation of Mc
Keywords:
Mass, element, compound, symbol, atoms, balanced
Balance the following equations:
C2H4 +
O2
CO2
CO2 + H2O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning