Chapter 4 https://ezto.mheducation.com/hm.tpx Be sure to answer all parts. Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate). Be sure to include states of matter. Note, salts of perchlorate are considered to be soluble. Part 1 out of 3 (a) Balance the equation: HBr(aq) + NH3(aq) 3HBR NH3 What is the ionic equation? What is the net ionic equation? Next part Check my work 3 amempts left eBook & Resources References Multipart Answer O Type here to search
Chapter 4 https://ezto.mheducation.com/hm.tpx Be sure to answer all parts. Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate). Be sure to include states of matter. Note, salts of perchlorate are considered to be soluble. Part 1 out of 3 (a) Balance the equation: HBr(aq) + NH3(aq) 3HBR NH3 What is the ionic equation? What is the net ionic equation? Next part Check my work 3 amempts left eBook & Resources References Multipart Answer O Type here to search
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter7: Reactions In Aqueous Solutions
Section: Chapter Questions
Problem 6CR: xplain to your friend what chemists mean by a precipitation reaction. What is the driving force in a...
Related questions
Question
Can you please answer the question and show the steps
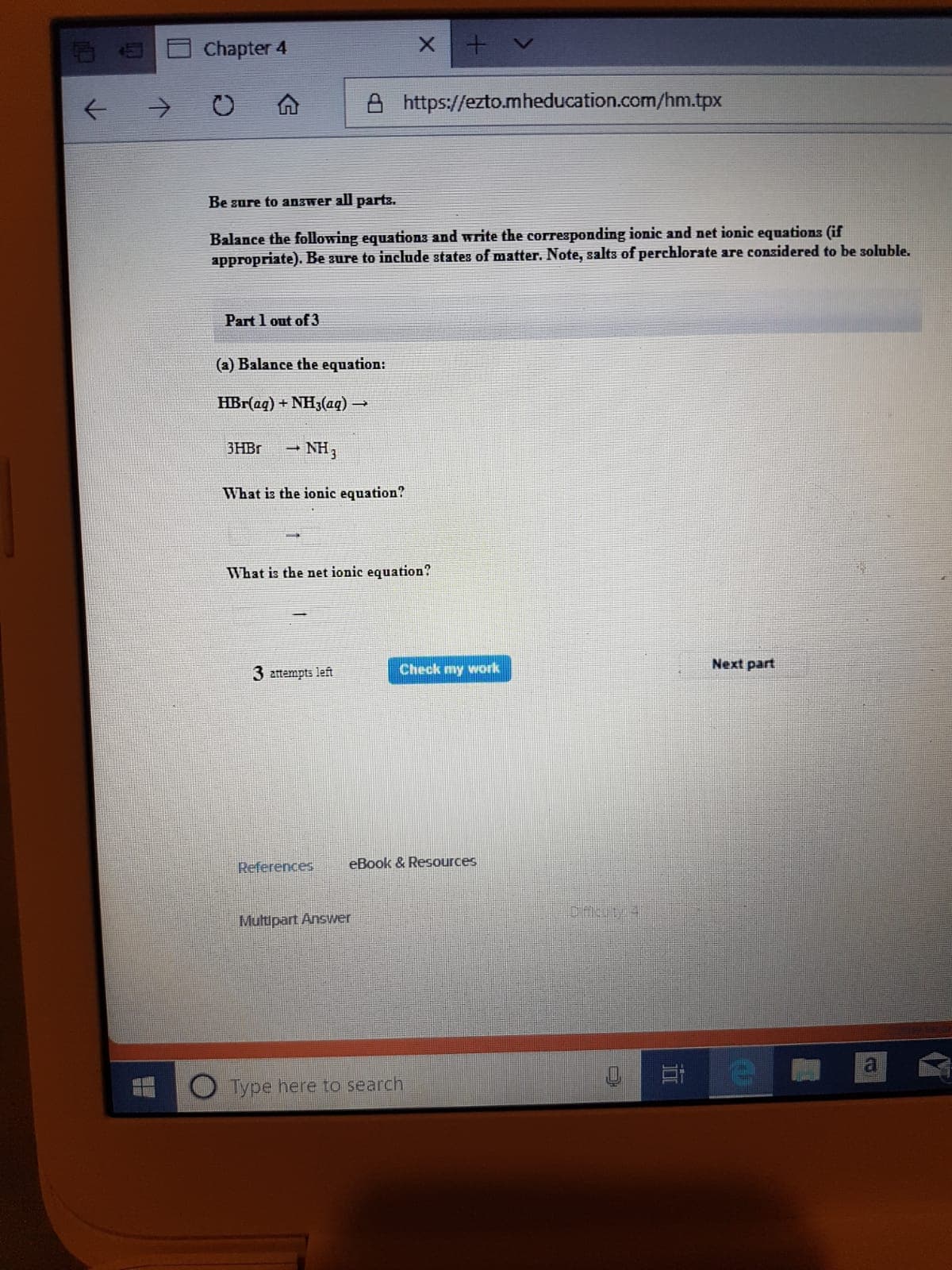
Transcribed Image Text:Chapter 4
https://ezto.mheducation.com/hm.tpx
Be sure to answer all parts.
Balance the following equations and write the corresponding ionic and net ionic equations (if
appropriate). Be sure to include states of matter. Note, salts of perchlorate are considered to be soluble.
Part 1 out of 3
(a) Balance the equation:
HBr(aq) + NH3(aq)
3HBR
NH3
What is the ionic equation?
What is the net ionic equation?
Next part
Check my work
3 amempts left
eBook & Resources
References
Multipart Answer
O Type here to search
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning