oman's Interacti X 68U9zhHgbWASuJSIN7n6mmMh30WJVXTzxEOQhl/edit tensions Help S 100 120 Spanish 24 and 2B Portfolio Infor X Calibri - 18 (a) on UA BIU + on + U (b) on t C 3 6. Explain why energy cannot be on the left side of an equation modeling nuclear fusion. 1 The following equation describes a typical fusion reaction that occurs in the sun. H + → He +n + energy Write the symbol for the missing reactant. 235U 235 U → 92 4 = Copy of Unit 7 Sample Work Which of these reactions successfully models a nuclear fission reaction? 239 + 239 U → 22U + energy 146 $7 ✔ → La + 20 « @ @ =- £ *- 5 87 + Br + 3n energy 6 What do nuclear fission, nuclear fusion, and radioactive decay have in common? How do they differ? 67°F
oman's Interacti X 68U9zhHgbWASuJSIN7n6mmMh30WJVXTzxEOQhl/edit tensions Help S 100 120 Spanish 24 and 2B Portfolio Infor X Calibri - 18 (a) on UA BIU + on + U (b) on t C 3 6. Explain why energy cannot be on the left side of an equation modeling nuclear fusion. 1 The following equation describes a typical fusion reaction that occurs in the sun. H + → He +n + energy Write the symbol for the missing reactant. 235U 235 U → 92 4 = Copy of Unit 7 Sample Work Which of these reactions successfully models a nuclear fission reaction? 239 + 239 U → 22U + energy 146 $7 ✔ → La + 20 « @ @ =- £ *- 5 87 + Br + 3n energy 6 What do nuclear fission, nuclear fusion, and radioactive decay have in common? How do they differ? 67°F
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter25: Nuclear Chemistry
Section: Chapter Questions
Problem 51GQ
Related questions
Question
Transcribed Image Text:oman's Interacti X
68U9zhHgbWASuJSIN7n6mmMh30WJVXTzxEOQhl/edit
tensions Help
S
100
120
Spanish 24 and 2B Portfolio Infor X
Calibri -
18
(a) on
UA
BIU
+
on + U
(b) on t
C
3
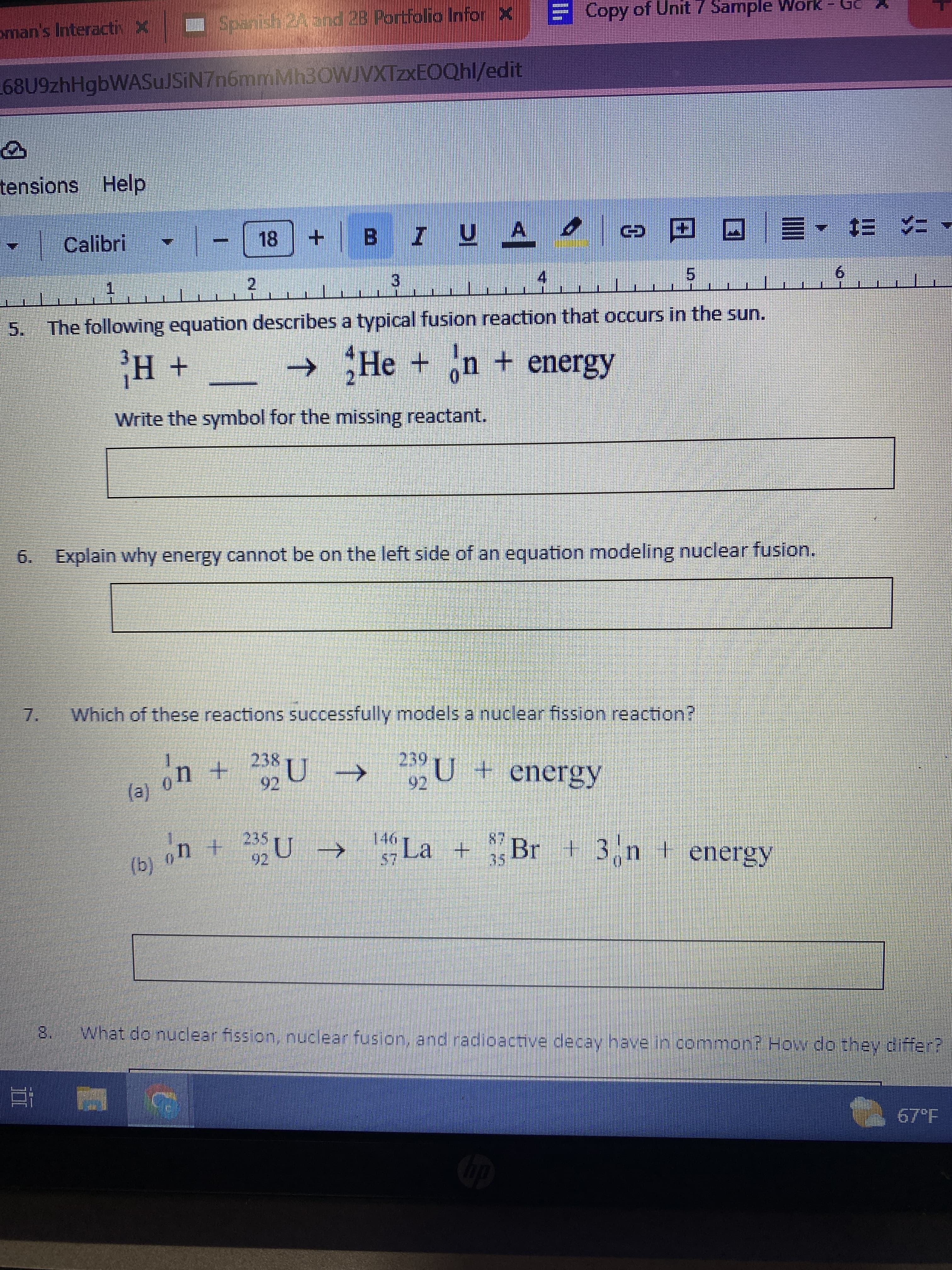
6. Explain why energy cannot be on the left side of an equation modeling nuclear fusion.
1
The following equation describes a typical fusion reaction that occurs in the sun.
H +
→ He +n + energy
Write the symbol for the missing reactant.
235U
235 U →
92
4
= Copy of Unit 7 Sample Work
Which of these reactions successfully models a nuclear fission reaction?
239
+ 239 U →
22U + energy
146
$7
✔
→ La +
20
« @ @ =- £ *-
5
87
+
Br + 3n energy
6
What do nuclear fission, nuclear fusion, and radioactive decay have in common? How do they differ?
67°F
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 11 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning