Choose the molecule(s) that will only show two signals, with an integration ratio of 2:3, in their 1H NMR spectum. Choosé one or more: O molecule A O molecule B O molecule C O molecule D O molecule E O molecule F O molecule G O molecule H
Choose the molecule(s) that will only show two signals, with an integration ratio of 2:3, in their 1H NMR spectum. Choosé one or more: O molecule A O molecule B O molecule C O molecule D O molecule E O molecule F O molecule G O molecule H
Chapter2: Life's Chemical Basis
Section: Chapter Questions
Problem 2SA: A molecule into which a radioisotope has been incorporated can be used as an ____________. a....
Related questions
Question
Transcribed Image Text:Choose the molecule(s) that will only show two signals, with an integration ratio of 2:3, in their 1H NMR spectum.
Choosé one or more:
O molecule A
O molecule B
O molecule C
O molecule D
O molecule E
O molecule F
O molecule G
O molecule H
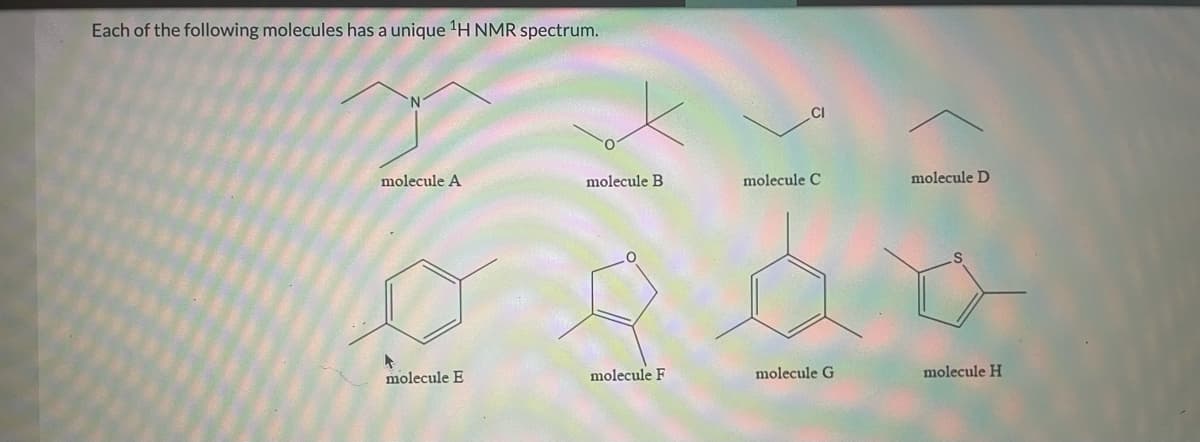
Transcribed Image Text:Each of the following molecules has a unique 'H NMR spectrum.
CI
molecule A
molecule B
molecule C
molecule D
molecule E
molecule F
molecule G
molecule H
Expert Solution
Step 1
The peak area of the 1H NMR spectrum is proportional to the amount of hydrogen present in a given chemical environment. As a result the ratio of these signals integration gives us the relative number of hydrogen in two peaks.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning


Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning