Class A 25 mL Pipette Calibration Actual Volume Mass of Water Delivered, g Calibration Initial Mass, g Final Mass, g #3 Delivered, mL 1 72.72 97.59 24.87 69.52 94.63 25.11 3 70.53 95.30 24.77
Class A 25 mL Pipette Calibration Actual Volume Mass of Water Delivered, g Calibration Initial Mass, g Final Mass, g #3 Delivered, mL 1 72.72 97.59 24.87 69.52 94.63 25.11 3 70.53 95.30 24.77
Chapter32: Gas Chromatography
Section: Chapter Questions
Problem 32.15QAP
Related questions
Question
Calculate actual volume delivered for #3
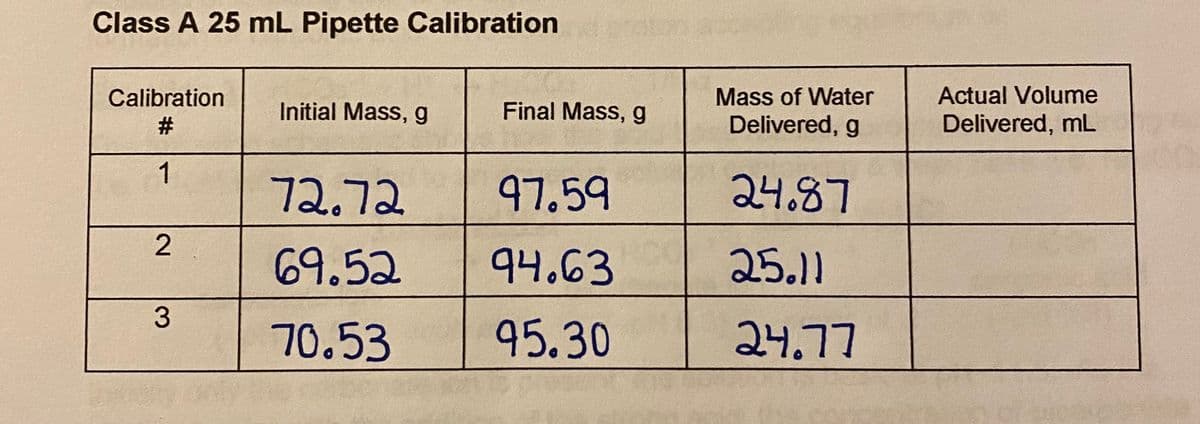
Transcribed Image Text:Class A 25 mL Pipette Calibration
Calibration
Mass of Water
Actual Volume
Initial Mass, g
Final Mass, g
Delivered, g
Delivered, mL
1
72.72
97.59
24.87
69.52
94.63
25.11
3
70.53
95.30
24.77
2.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning
