Complete the chart by determining the state of matter (solid, liquid, or gas) of each of the compounds at room temperature (20 °C). Drag the appropriate labels to their respective targets. > View Available Hint(s) Reset Help Properties of water compared to other low-molecular weight compounds Compound Molar Mass Melting Dielectric point (°C) constant Boiling State at point (°C) room (g/mol) temperature solid CH4 16.0 -182 -162 1.7 liquid liquid NH3 17.0 -78 -33 17 gas gas H2O 18.0 +100 78 solid HF 20.0 -83.1 19 17 solid
Complete the chart by determining the state of matter (solid, liquid, or gas) of each of the compounds at room temperature (20 °C). Drag the appropriate labels to their respective targets. > View Available Hint(s) Reset Help Properties of water compared to other low-molecular weight compounds Compound Molar Mass Melting Dielectric point (°C) constant Boiling State at point (°C) room (g/mol) temperature solid CH4 16.0 -182 -162 1.7 liquid liquid NH3 17.0 -78 -33 17 gas gas H2O 18.0 +100 78 solid HF 20.0 -83.1 19 17 solid
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter5: Alkenes: Bonding, Nomenclature, And Properties
Section: Chapter Questions
Problem 5.31P
Related questions
Question
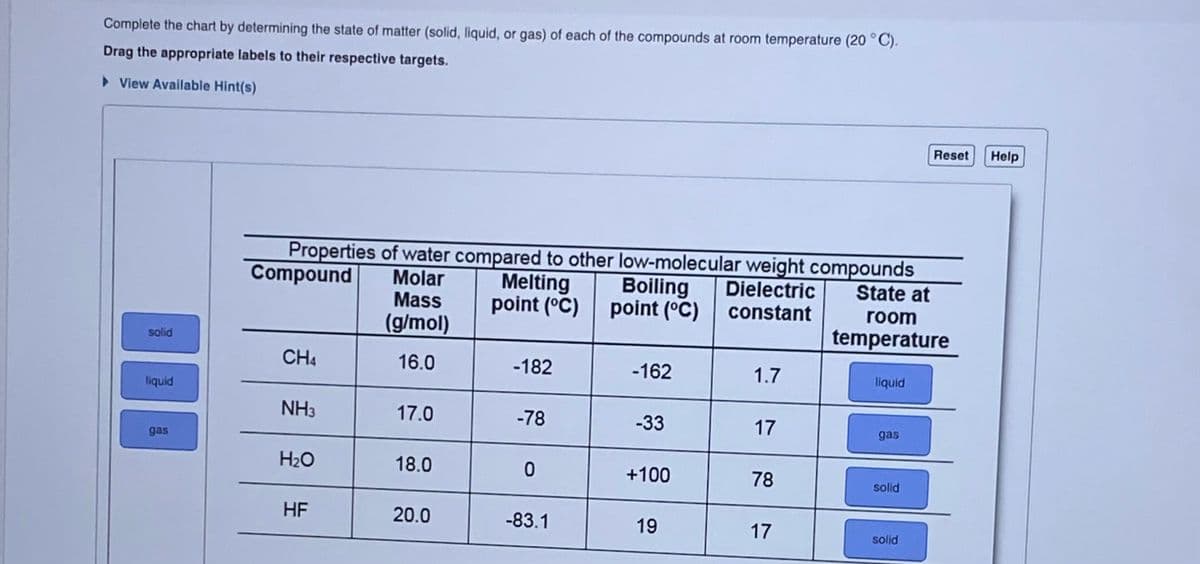
Transcribed Image Text:Complete the chart by determining the state of matter (solid, liquid, or gas) of each of the compounds at room temperature (20 °C).
Drag the appropriate labels to their respective targets.
> View Available Hint(s)
Reset
Help
Properties of water compared to other low-molecular weight compounds
Compound
Molar
Mass
Melting
Boiling
Dielectric
constant
State at
point (°C) point (°C)
room
(g/mol)
temperature
solid
CH4
16.0
-182
-162
1.7
liquid
liquid
NH3
17.0
-78
-33
17
gas
gas
H2O
18.0
+100
78
solld
HF
20.0
-83.1
19
17
solid
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning