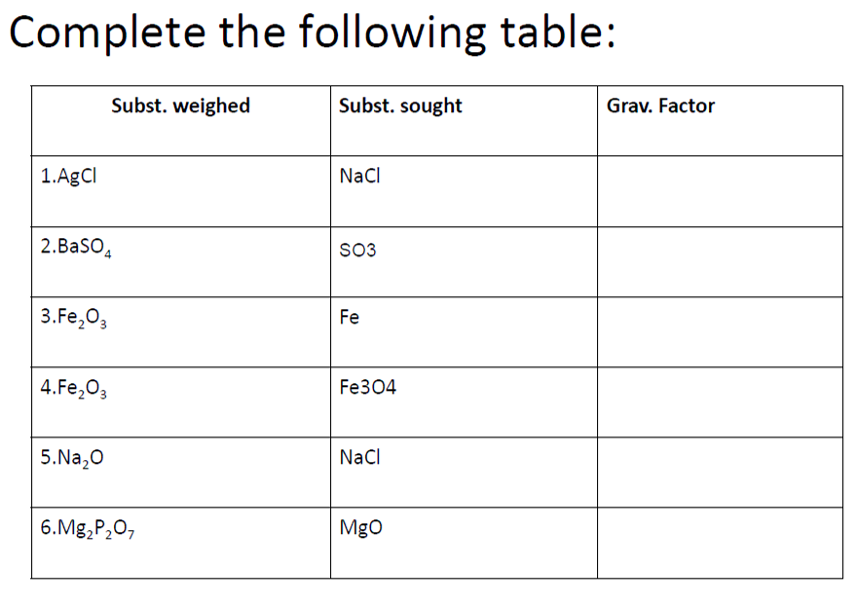
Complete the following table: Subst. weighed Subst. sought Grav. Factor 1.AgCI NaCl 2.Baso, SO3 3.Fe,03 Fe 4.Fe,03 Fe304 5.Na,0 NaCl 6.Mg,P2O, Mgo
Q: Determination of AHf of NH4CI(s) & AH soln AHf of NH4CI(s) using your values of AH neut AHf of…
A: Please note that dissolution is an endothermic process. So, ∆Hsol should be 8.68 kJ/mol ( a positive…
Q: Siven reaction is carried out in acidic medium with a yield of 91.5%. How many grams of CH,CHO is…
A:
Q: How is the molarity of a 0.50 M KBr solution changed when water is addedto double its volume?
A: Molarity is used in the calculation of quantity of solute or the volume of solvent in a solution,…
Q: Brand of Sample: Trial 1 Volume of sample 10.00 mL 25.63 ml Final reading - standard NAOH solution…
A: Vinegar is a dilute solution of acetic acid. It is used as salad dressing and preservative. The data…
Q: Based on your ICE table and definition of Ka, set up the expression for Ka in order to determine the…
A: As CH3COOH is a weak acid, it does not completely dissociate into its ions, CH3COO- and H+. Let x M…
Q: A 15.0% by weight solution was prepared using 90.0g of KCl and the resulting density of the solution…
A: Given % by weight = 15 % Mass of solute = 90.0 g Density of solution = 1.101 g/ml
Q: 0.119 (measured) 3) Molarity of final unknown solution (calculated from calibration curve) M.
A: The slope-intercept form of line is y = mx + c, here, m = slope of the line and c = y-intercept. The…
Q: Before investigating the scene, the technician must dilute the luminol solution to a concentration…
A:
Q: 2. Given the following information: is there any significant difference between the 2 methods?…
A: F-test is used to compare the results of two different methods.
Q: 7. Compute for the % mass or weight salt solution by mixing 622g of NaCl with 1.86 liters of H20.…
A:
Q: How many milliliters of 9.00 % stock NaCl solution are needed to create 25.00 mL of 0.900 % (m/v)…
A: C1V1 = C2V2 as number of moles remain same after dilution .or we can say amount of solute remain…
Q: Calculate the equivalent weight of KMnO₄ in the following reaction: MnO₄⁻ + CN⁻ + H₂O ⟶ MnO₂ (s) +…
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: When working with very low concentrations, such as 0.000245 M, it can be more convenient to…
A: Molarity: It is the number of moles of solute present per Litre of the solution. The units of…
Q: 21.929 Mass empty test tube and stopper Mass test tube, stopper and cyclohexanc Mass of cyclohexane…
A: Mass of cyclohexane(Solvent) = 6.39 g Mass of unknown in mixture 1 = 0.06 g' Mass of unknown in…
Q: How do I calculate question 1 part b (in the picture)? I don’t even know where to start. Thank…
A: The Solubility v/s temperature data for an organic substance A dissolved in water is given. Now,…
Q: A mixer containing 100 gal of Na,SO, solution (4 Ibs of Na2S04 per gal) is attached to a reservoir…
A:
Q: B) Analysis of unknown aspirin tablet 1.Mass of aspirin tablet= 2.Concentration of ASA unknown from…
A: First, we have to draw the absorbance versus concentration(M) plot. From the linear regression…
Q: Using a 10mL pipette, you prepare a standard solution by pipetting 10.00mL of the standard iron…
A: A particular pipette has an approximate tolerance or uncertainty value. Based on these uncertainty…
Q: i cant seem to get the right answer for this, could you please explain how i use the constant? like…
A:
Q: Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. From the stock solution (0.020…
A:
Q: What are thier balanced equation (with phases) and what happens when they are mixed? Part 1: a.)…
A:
Q: 1) Mass of aspirin used (g) 0.088g 2) Absorbance for final unknown solution (measured) 3) Molarity…
A: Aspirin is a drug used for controlling pain, reducing fever etc. Aspirin tablet is not made up of…
Q: Which two statements are correct? a) The normality of the K2Cr2O7 solution is 0.41 N b) The…
A: When dichromate ion in acidic medium, treated with ferrous ion, then Ferric ion and chromium (III)…
Q: Mass Na CO3 Vi HC1,mL 0.00 0.10 Vf HC1,mL 36.75 Trial VHC) used, ml MHCI 1 0.2135 0.2025 0.2112…
A: Since you have asked a question with multiple sub-parts, as per our company guidelines we are…
Q: Calculate the gravimetric factor of: FeO in Fe2O3 Bi2S3 in BaSO4 BaSO4 in (NH4)2SO4 Mn2O3 in Mn3O4…
A:
Q: Data Sheet II. Delivering Solution from Your Buret 17.28 (1) final buret reading, mL (2) initial…
A:
Q: STANDARDIZE THE Na,$;O3.5H;O WITH KIO,. Weight of KIO, = 0.5325 g V of KIO, = 250 mL N KIO, V KIO,…
A: Given:
Q: : A 12.1 Moles NaOH solution means there are 12.1 moles of NaOH in liter of solution. Preparation of…
A:
Q: Will Ag2 C03 (Ksp = 8.1 250.0ml of 0.0062 m 0.00014 m Na2 Co3 are
A: Solubility equilibrium of Ag2CO3 Ag2CO3(s) ----> 2Ag+(aq.) + CO32- Qsp = [Ag+]2[CO32-] =…
Q: 3. (a) If 10.0 mL of H;SO4 (sp. Gr. 1.50, containing 48.7% of combined SO3 by weight) is diluted to…
A: 3a) From the given data - 48.7 g of SO3 is present in 100 g of total solution. 10.0 mL…
Q: M = wt (g) 1000 M. wt (B v (mL) mol 0.250 M: wt (g) 1000 58.4 () 1 (mL) wt (g) 0.0146 B mL %3D…
A:
Q: A. Standardization of NaOH Solution TRIAL [HCI] Volume Moles Moles Volume [NAOH] HCI НОEN 25.75 IOH…
A:
Q: #13 Mass percent of asprin in tablet
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: H. HyC-NH, H, H,0 N.
A:
Q: of (a) molarity ? erum? (C) ppm by wt? 2. How many mL of a 0.250 M KA solution must be diluted to…
A:
Q: Commercial perchloric acid (100.46 g/mol) 71% (w/w) with specific gravity of was diluted by taking…
A: Given: %(w/w) of perchloric acid i.e. HClO4 = 71 %. Volume of HClO4 solution taken = 15.00 mL…
Q: 4. Calculate the mass of water evaporated from the solution? 0.0414 M 5. Calculate the density of…
A: 4. The mass of water evaporated from solution is determined by considering mass of CuSO4 solution…
Q: Ingliest mg po H. 3. H-C-O-C-H .. a. H. H H H-C-C-ÖH b. H H…
A: Boiling point of alcohol is always greater than ethers, aldehydes or alkanes having similar…
Q: d. Estimate the Ka of kobeicin using the data from the second derivative plot. e. A separate…
A: Given data:
Q: A 25mL volumetric pipet is used to deliver a sample of the stock solution marked “0.6000M X2SO4”…
A: Given : concentration of X2SO4 stock solution = 0.6000 M Volume of stock solution used = 25 mL And…
Q: Please help me to answer a question. I have 2 mg/mL stock solution and I need to make standards of…
A: Concentration is defined as amount of solute per unit amount of solution. Dilution refers to…
Q: A 0.53 g sample containing KBr (MW=119) is dissolved in 50 mL of distilled water. Titrating with…
A:
Q: A. Standardizing a Dilute Solution of Sodium Hydroxide, NaOH (aq) КНС,Н,О, (аq) + NaOH (aq) H,O (1)…
A: The reaction is standarization of NaOH With respect of KHP.
Q: 100 ml g 0:4 Nop HSq mixed with 100m) oro.2 N N<OH, Ihen Final Normaliliy og Resxelt toletian is A)…
A:
Q: Calculate the Standardized Molarity of NaOH via dimensional analysis. Show your process. Use this…
A:
Q: I each result so that it con solute standard deviations.
A: Given as, y=1.203±0.004×103 +1.825±0.005×102 -34.012±0.0013.9201±0.0006×10-32
Q: NAOH (ag) + HCI NaCl (ag) + H20 (1) (aq) -> 31.88 mL of 0.09951 M NaOH was used to titrate 7.00 mL…
A: We will calculate moles of NaOH , then moles of HCl and at last molarity of HCl.
Q: Concentration Initial Final Volume Mass percent of acetic acid of Heinz volume Volume(mL) (mL) of in…
A:
Q: Cat Noir was tasked to determine the concentration of a CuSO4 unknown solution. She prepared 5…
A: Lambert Beer's Law depicts that when a monochromatic light flows through an adsorbing medium, the…
Q: In this station, you will find a sample of table salt; the chemical formula of table salt is NaCl.…
A: Formula to calculate the mass percent of solute WV in solution. Mass percentage of solute WV=Mass of…
Kindly provide solutions in completing this table (finding gravimetric factor) since they are all connected. Pls skip if unsure or not willing to show solutions under 1-6. Thank you! [p.s. kinda confused with how to treat the two columns especially subst. sought]
Step by step
Solved in 3 steps with 2 images

- Write the empirical formula for the hydrated KAl(SO4)2, based on moles of anhydrous KSI(SO4)2= 0.046 mol molar mass of H2O= 18g/ mol moles of H2O= 0.0444mol ratio pf moles H20 to moles of anhydrous KAI(SO4)2= 9.65/1 . Show all work including units. Hint: if the ratio of moles of H2O to moles of anhydrous KAl(SO4)2 was 4, then the empirical formula would be: KAl(SO4)2•4H2O.Determination of the amount of water, mass of crucible + dehydrated sample 16.2265g, mass of dehydrated Sample is 1.3031g, mass of water is 0.3587g, moles of water is 0.01991moles,DETERMINATION of the amount of copper, mass of copper 0.6291g, moles of copper 9.900 × 10 to the negative 3 moles, Determination of the amount of chlorine, calculate mass of copper + water.The hydrated salt, MY3·xH2O, was heated strongly in a crucible to remove all the watermolecules of crystallization.The data are for this experiment are given in the table below.Mass of empty crucible and cover 29.73 gMass of crucible, cover, and hydrated salt 32.72 gMass of crucible, cover, and anhydrous salt 31.52 gMolar mass of anhydrous salt 241.88 g/mol(i) Calculate the % composition by mass of water in the hydrated salt.(ii) Determine the value of x.(iii) M is in Period 4 of the Periodic Table of the Elements.If Y = NO3–, identify the element M.(iv) What is the name of this hydrated salt? ___
- a hydrated CuSO4 salt was heated and thus dehydrated. Determine the % H2O by weight in the hydrated CuSO4 salt. SHOW WORK TO RECEIVE FULL CREDIT mass of empty dish 43.571 gmass of dish + hydrated salt 44.326 gmass of dish + dehydrated salt 44.054 g 4. Using the data found in question number 3, determine the empirical formula for the hydrated CuSO4salt. What is the chemical name of this dehydrated salt? 5. Suppose that after your second heating, you were impatient and did not wait until your porcelain evaporating dish to cool. Instead, you weighed it while it was still hot. How would this specifically affect the calculated percentage of water in the compound?KSCN(aq) + Ag+(aq) → AgSCN(aq) + K+(aq) By calculation she determines the buret delivered: 6.52108 moles of KSCN. The moles of Ag+ which were present in her sample was:Could you help me this calculation part? Mass of empty, dry crucible (g): 0.5gMass of crucible with sodium bicarbonate (g): 0.5gMass after first heating (g): 0.5gMass after second heating (g): XgMass after third heating (if needed) (g): YgC a l c u l a t i o n s : Show calculations and include all units. Circle your final answers, and include them in the data section as needed. 1. Calculate the mass of sodium bicarbonate sample used: 2. Calculate the theoretical yield of sodium carbonate based on your starting mass of sodium bicarbonate: 3. Calculate the mass of sodium carbonate obtained (this is your experimental yield): 4. Calculate the percent yield for sodium carbonate: Experimental x 100 = Theoretical
- A mixture weighing 28.516g containers 4.518g NH4CL, 15.20g SiO2, with the remainder NaCl. find the percent, weight, of each component. Show you method for making the calculations.Mass of empty beaker=16.26Volume of CuSO4 5H2O=30mLMolarity of CuSO4 5H2O used=0.542Moles of CuSO4 5H2O=0.016Mass of aluminum foil used=0.26gMass of beaker and copper product=52.12gMass of copper metal product=35.86gMoles of Al used = 0.009636 molMoles of CuSO4 5H2O used=0.016mol Find the moles of copper product based on moles of Al.Find the moles of copper product based on moles of CuSO4 5H2OFind the limiting reactant in gramsGiven Active Ingredient: precipitated sulfur (ointment) Raw Materials: 500 g calcium polysulphide and 1.5 kg hydrochloric acid Actual Yield: 343.4g precipitated sulfur Formulation: 250 mg per jar Dosage form: Ointment packaging:100 jars per box Synthesis and Packaging (Need answer)- Balanced Chemical Equation:- % composition by mass of each compound:- Mass to Mass Stoichiometry Calculation:- Limiting Reagent:- Excess Reagent:- Amount (g) in excess: % Yield:- Number of dosage form and packaging that can be produced from stoichiometric solution:
- Balance each of the following equations. (State indicators are not required) Please answer (a) through (o). (a) Cdf2 + NaBr --> CdBr2 + NaF (b) Cr + F2 --> CrF3 (c) Ca + H2O --> Ca(OH)2 + H2 (d) Bi(NO3)3 + Na2S --> Bi2S3 + NaNO3 (e) C2H5OH + O2 --> CO2 + H2O (f) V + S8 --> V2S5 (g) LiNO3 + Li --> Li2O + N2 (h) Ca3(PO4)2 + H2SO --> CaSO4 + H3PO4 (i) PH3 + O2 --> P2O5 + H2O (j) Ba + Ag3PO4 --> Ba3(PO4)2 + Ag (k) Ca(Clo3)2 --> CaCl2 + O2 (l) C12H22O11 + O2 --> CO2 + H2O (m) Ca2C + H2O --> Ca(OH)2 +CH4 (n) NH4Br + BaO --> NH3 + BaBr2 + H2O (o) LiAlH4 + BF3 --> LiF + AlF3 + B2H6For the following table fill in the missing values and calculate the theoretical yield. A + 2B à C moles 0.00242 0.00242 ?_________ MW ?_________ 98.06 164.16 g ?_________ ?__0.00002467_______ ?_________ mL 0.20 d(g/mL) 0.80Be sure to answer all parts. Fluorite, a mineral of calcium, is a compound of the metal with fluorine. Analysis shows that a 36.77−g sample of fluorite contains 17.87 g of fluorine. Calculate the following: (a) Mass of calcium in the sample. _________g Ca (b) Mass fractions of calcium and fluorine in fluorite. Calculate to 3 significant figures. mass fraction Ca mass fraction F (c) Mass percents of calcium and fluorine in fluorite. Calculate to 3 significant figures. mass % Ca mass % F

