Concentration of standardized HCl solution (M) 0.1563 Sample 1 Sample 2 Sample 3 Temperature of sample (°C) 0.60 16.00 25.00 Volume of sample analyzed (mL) 5.00 5.00 5.00 Initial buret reading (mL) 24.60 19.20 8.20 Final buret reading (mL) 29.53 28.00 20.76
Concentration of standardized HCl solution (M) 0.1563 Sample 1 Sample 2 Sample 3 Temperature of sample (°C) 0.60 16.00 25.00 Volume of sample analyzed (mL) 5.00 5.00 5.00 Initial buret reading (mL) 24.60 19.20 8.20 Final buret reading (mL) 29.53 28.00 20.76
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter13: Solutions And Their Behavior
Section13.5: Colloids
Problem 1.2ACP: How many theoretical plates are required to produce a solution with a mole fraction of hexane...
Related questions
Question
Calculate the molar solubility and Ksp of borax at each of the measured temperatures.
Equations
Na2B4O5(OH)4·8H2O(s) ⇌ 2Na+(aq) + B4O5(OH)42-(aq) + 8H2O(l)
B4O5 5(OH)42-(aq) + 2H+(aq) + 3H2O(l) → 4H3BO3(aq)
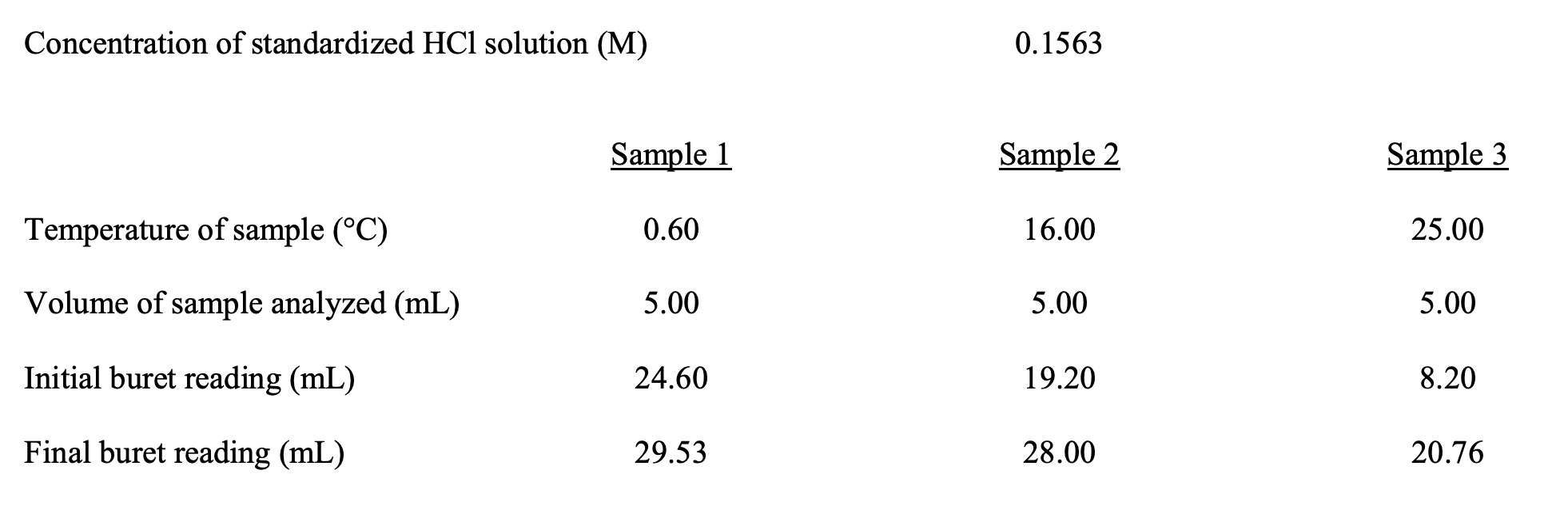
Transcribed Image Text:Concentration of standardized HCl solution (M)
0.1563
Sample 1
Sample 2
Sample 3
Temperature of sample (°C)
0.60
16.00
25.00
Volume of sample analyzed (mL)
5.00
5.00
5.00
Initial buret reading (mL)
24.60
19.20
8.20
Final buret reading (mL)
29.53
28.00
20.76
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning