Consider taking the Legendre transform of the energy, E(S,V,N), with respect to S, V, and N, to get a new thermodynamic potential, X(T.p,u). (a) Show that X(T.p,µ) is identically zero. (b) What does this imply about the variables T, p, and µ? Where have you already seen this result before?
Consider taking the Legendre transform of the energy, E(S,V,N), with respect to S, V, and N, to get a new thermodynamic potential, X(T.p,u). (a) Show that X(T.p,µ) is identically zero. (b) What does this imply about the variables T, p, and µ? Where have you already seen this result before?
Related questions
Question
100%
It's a
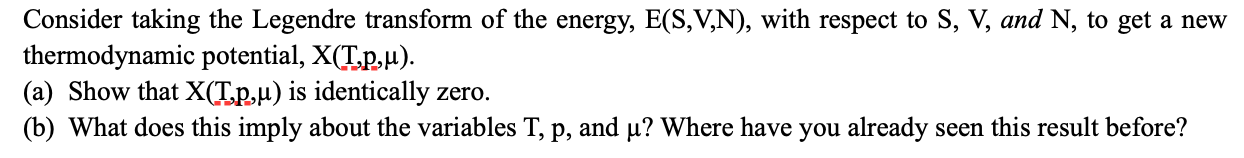
Transcribed Image Text:Consider taking the Legendre transform of the energy, E(S,V,N), with respect to S, V, and N, to get a new
thermodynamic potential, X(T.p,u).
(a) Show that X(T.p,µ) is identically zero.
(b) What does this imply about the variables T, p, and µ? Where have you already seen this result before?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 8 steps with 8 images
