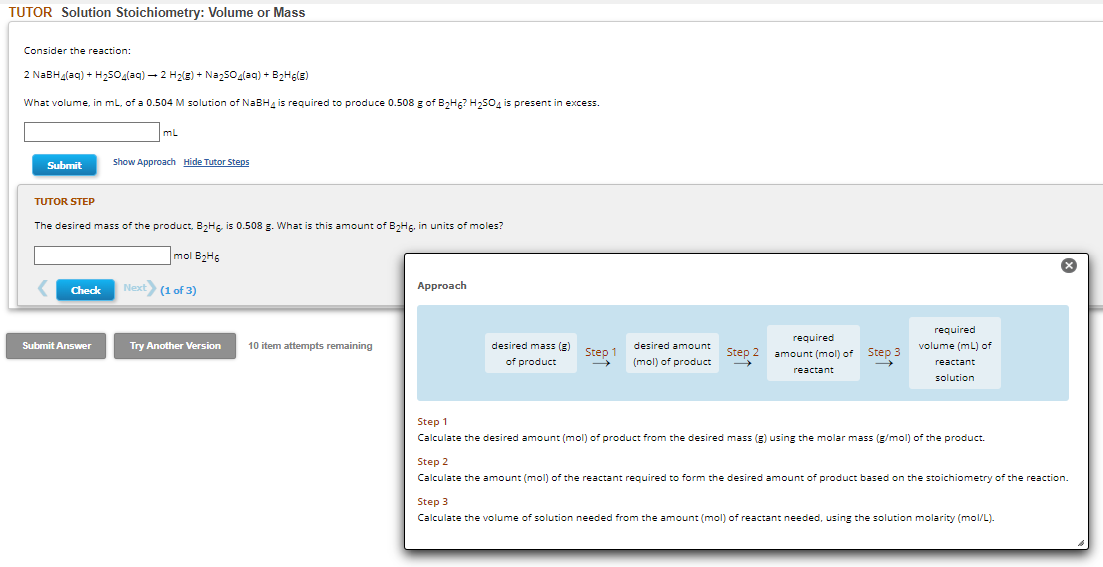
Consider the reaction: 2 NaBH₂(aq) + H₂SO4(aq) → 2 H₂(g) + Na₂SO4(aq) + B₂H6(E) What volume, in mL, of a 0.504 M solution of NaBH4 is required to produce 0.508 g of B₂H6? H₂SO4 is present in excess. Submit mL Check Show Approach Hide Tutor Steps TUTOR STEP The desired mass of the product. B₂H6, is 0.508 g. What is this amount of B₂H, in units of moles? mol B₂H6 Next> (1 of 3) Approach
Consider the reaction: 2 NaBH₂(aq) + H₂SO4(aq) → 2 H₂(g) + Na₂SO4(aq) + B₂H6(E) What volume, in mL, of a 0.504 M solution of NaBH4 is required to produce 0.508 g of B₂H6? H₂SO4 is present in excess. Submit mL Check Show Approach Hide Tutor Steps TUTOR STEP The desired mass of the product. B₂H6, is 0.508 g. What is this amount of B₂H, in units of moles? mol B₂H6 Next> (1 of 3) Approach
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter10: Gases And Their Properties
Section: Chapter Questions
Problem 100IL: You are given a solid mixture of NaNO2 and NaCl and are asked to analyze it for the amount of NaNO2...
Related questions
Question
Transcribed Image Text:TUTOR Solution Stoichiometry: Volume or Mass
Consider the reaction:
2 NaBH₂(aq) + H₂SO4(aq) → 2 H₂(g) + Na₂SO4(aq) + B₂H6(E)
What volume, in mL, of a 0.504 M solution of NaBH4 is required produce 0.508 g of B₂H6? H₂SO4 is present in excess.
Submit
TUTOR STEP
Check
mL
The desired mass of the product. B₂H, is 0.508 g. What is this amount of B₂H6, in units of moles?
mol B₂H6
Submit Answer
Show Approach Hide Tutor Steps
Next (1 of 3)
Try Another Version
10 item attempts remaining
Approach
desired mass (g)
of product
Step 1
desired amount
(mol) of product
Step 2
→
required
amount (mol) of
reactant
Step 3
required
volume (mL) of
reactant
solution
Step 1
Calculate the desired amount (mol) of product from the desired mass (g) using the molar mass (g/mol) of the product.
Xx
Step 2
Calculate the amount (mol) of the reactant required to form the desired amount of product based on the stoichiometry of the reaction.
Step 3
Calculate the volume of solution needed from the amount (mol) of reactant needed, using the solution molarity (mol/L).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning