Consider the remperagture composition diagram showe bolow for bexane and heptane For he mituRe of heann 1724 g (MW6.18 gimo end ttanes 30 0 MW-10021 gmal what temperatune wil the muture begjin to hal and what is the boling point of the liquid formed by condensation of the vapo? Temperature - composition diagram for a binary miture of hexane (bp= 63 "C) and heptane (bp-90 "C) 90 90 80 70 70 60 60 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 Mole fraction of heptane Temperature, "C
Consider the remperagture composition diagram showe bolow for bexane and heptane For he mituRe of heann 1724 g (MW6.18 gimo end ttanes 30 0 MW-10021 gmal what temperatune wil the muture begjin to hal and what is the boling point of the liquid formed by condensation of the vapo? Temperature - composition diagram for a binary miture of hexane (bp= 63 "C) and heptane (bp-90 "C) 90 90 80 70 70 60 60 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 Mole fraction of heptane Temperature, "C
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.19QAP
Related questions
Question
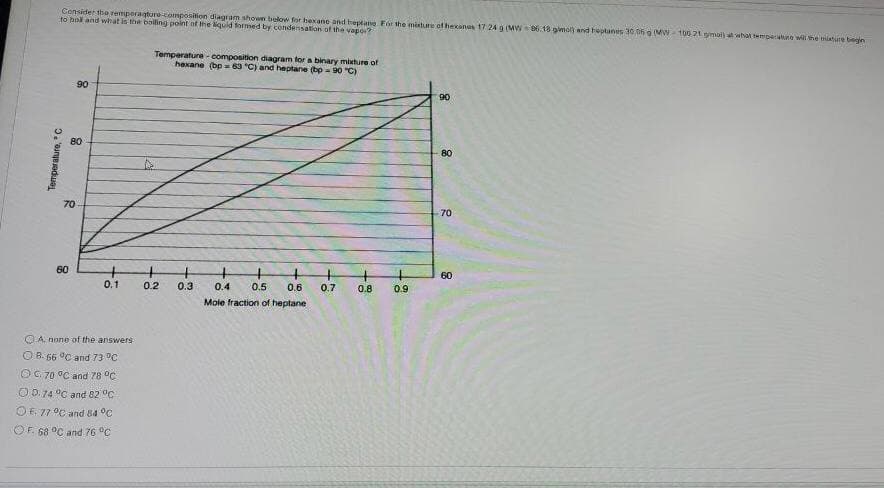
Transcribed Image Text:Censider the remperagture composition diagram shown below for bexane and heptane For he misture of heanen 17 24 9 (MW6.18 gimol and heptanes 30 06 o (MW100.21 gmai what tempeatine wili the miature begn
to hol and what is the boliling point of the liquid Sormed by condensation of the vapo?
Temperature - composition diagram for a binary miehure of
haxane (bp= 63 "C) and heptane (bp - 90 "C)
90
90
80
80
70-
70
60
60
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
Mole fraction of heptane
CLA. none of the answers
O B. 66 °C and 73 °C
OC. 70 °C and 78 °C
O D.74 °C and 82 °C
O E. 77 °C and 84 °C
OF 68 °C and 76 °C
Temperature, "C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole