Consider the titration of 24.0 mL of 0.200 M Na A (a weak base) and 0.100 MH CI. Calculate the pH of the solution after 9.0 mL of H CI has been added. The Kb of the base is 1.93E-5 To avoid rounding errors report your answer with two decimal places. Your Answer:
Consider the titration of 24.0 mL of 0.200 M Na A (a weak base) and 0.100 MH CI. Calculate the pH of the solution after 9.0 mL of H CI has been added. The Kb of the base is 1.93E-5 To avoid rounding errors report your answer with two decimal places. Your Answer:
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter17: Principles Of Chemical Reactivity: Other Aspects Of Aqueous Equilibria
Section17.3: Acid-base Titrations
Problem 17.6CYU: The titration of 0.100 M acetic acid with 0.100 M NaOH is described in the text. What is the pH of...
Related questions
Question

Transcribed Image Text:Consider the titration of 24.0 mL of 0.200 M Na A (a weak base) and 0.100 MH CI.
Calculate the pH of the solution after 9.0 mL of H CI has been added. The Kb of the
base is 1.93E-5
To avoid rounding errors report your answer with two decimal places.
Your Answer:
Expert Solution
Step 1
No of moles of weak base, NaA = Molarity × Volume = 0.200M×24.0mL = 4.80 mmoles
No of moles of strong acid, HCl = Molarity × Volume = 0.100M×9.0mL = 0.90 mmoles
Step 2
When NaA and HCl are titrated, the net ionic equation is:
The equilibrium moles can be determined from ICE table as follows:
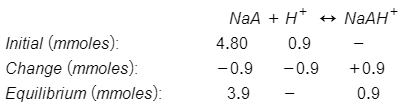
Total volume = 24.0mL + 9.0mL = 33.0 mL
Thus, [NaA] = 3.9mmoles/33.0mL
[NaAH+] = 0.9mmoles/33.0mL
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning