Constants I Peri Part C Suppose you have 3.50x10 8 reams of lead (Pb). How many lead atoms do you have? Express the number of atoms numerically. View Available Hint(s) VOAD ? atoms Previous Answers Submit X Incorrect; Try Again; 4 attempts remaining Next Provide Feedback MacBook Air DII DD F6 F7 F12 F8 F10 F11 F9 & + t
Constants I Peri Part C Suppose you have 3.50x10 8 reams of lead (Pb). How many lead atoms do you have? Express the number of atoms numerically. View Available Hint(s) VOAD ? atoms Previous Answers Submit X Incorrect; Try Again; 4 attempts remaining Next Provide Feedback MacBook Air DII DD F6 F7 F12 F8 F10 F11 F9 & + t
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.19QAP
Related questions
Question
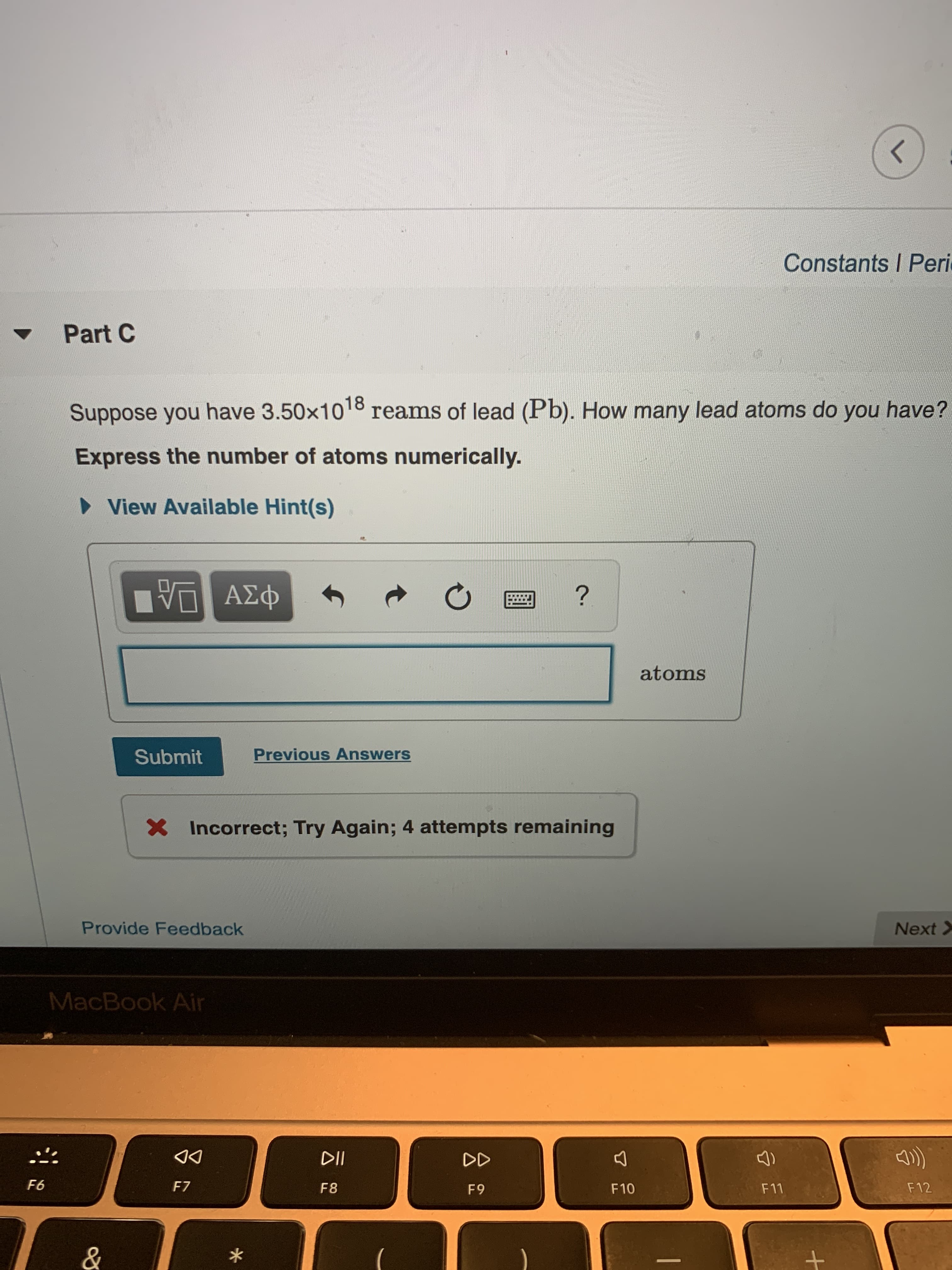
Transcribed Image Text:Constants I Peri
Part C
Suppose you have 3.50x10 8 reams of lead (Pb). How many lead atoms do you have?
Express the number of atoms numerically.
View Available Hint(s)
VOAD
?
atoms
Previous Answers
Submit
X Incorrect; Try Again; 4 attempts remaining
Next
Provide Feedback
MacBook Air
DII
DD
F6
F7
F12
F8
F10
F11
F9
&
+
t
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you



