Chapter6: The States Of Matter
Section: Chapter Questions
Problem 6.9E
Related questions
Question
Transcribed Image Text:app.101edu.co
Apps M Gmail
YouTube
金News
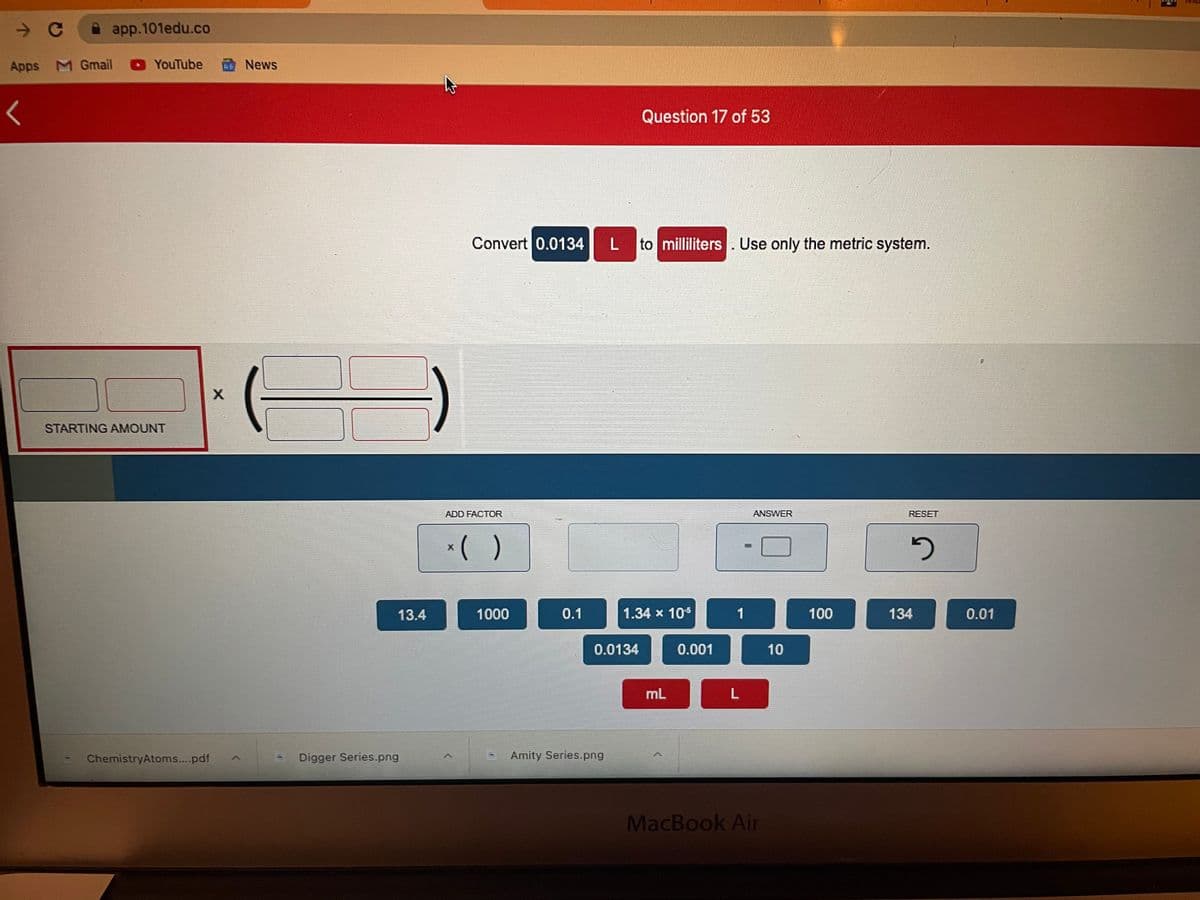
Question 17 of 53
Convert 0.0134
to milliliters. Use only the metric system.
STARTING AMOUNT
ADD FACTOR
ANSWER
RESET
*( )
13.4
1000
0.1
1.34 x 105
1
100
134
0.01
0.0134
0.001
10
mL
ChemistryAtoms....pdf
Digger Series.png
Amity Series.png
MacBook Air
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

