ct.mheducation.com t # 2 6 Saved Select all that apply. Select the compounds that are more soluble in acidic solution than in pure water: Ca(OH)2 Mgą(PO)2 O PbBr, < Prev 3 of 5 Next >
ct.mheducation.com t # 2 6 Saved Select all that apply. Select the compounds that are more soluble in acidic solution than in pure water: Ca(OH)2 Mgą(PO)2 O PbBr, < Prev 3 of 5 Next >
Chapter16: Applications Of Neutralization Titrations
Section: Chapter Questions
Problem 16.21QAP
Related questions
Question
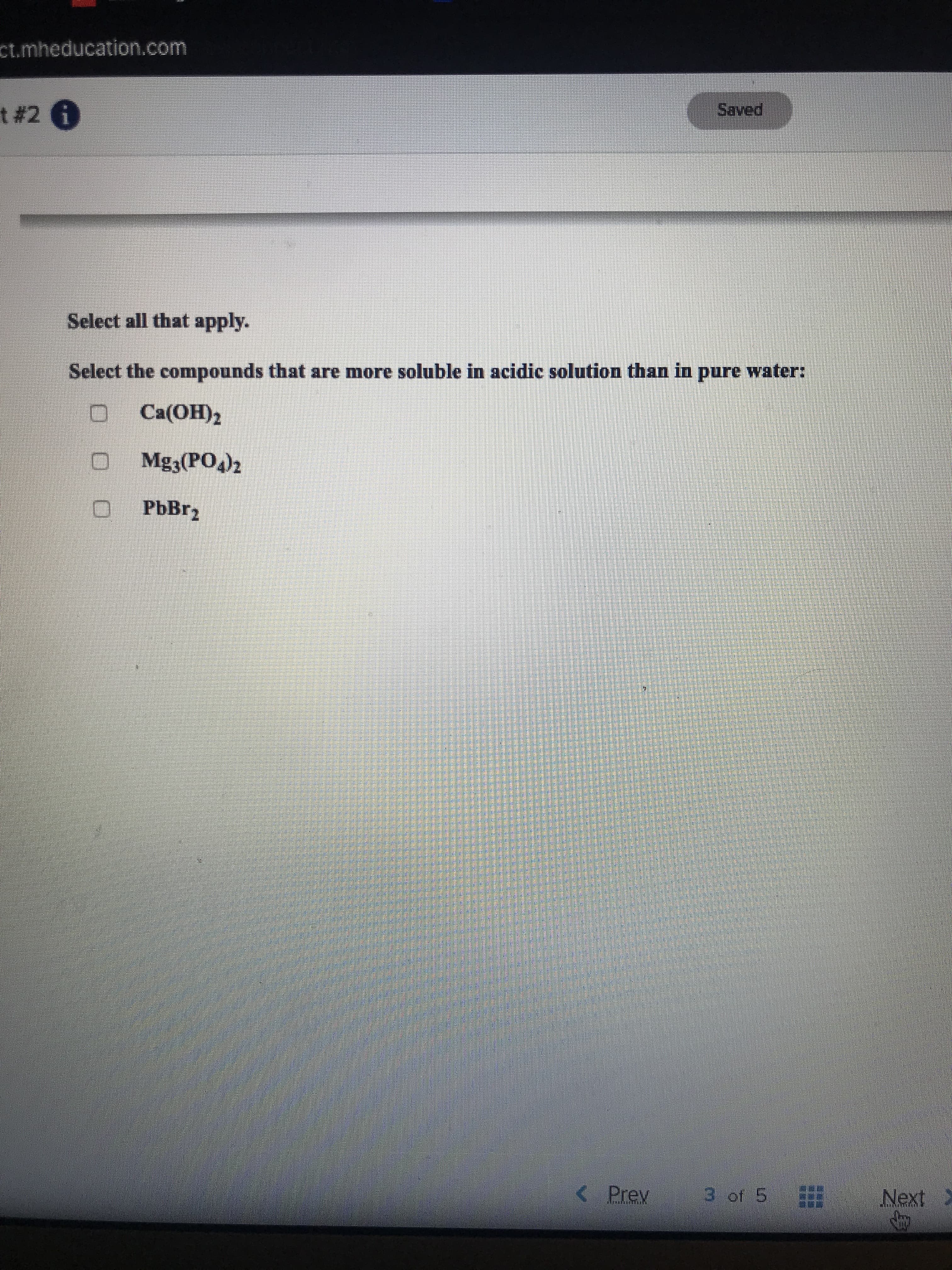
Transcribed Image Text:ct.mheducation.com
t # 2 6
Saved
Select all that apply.
Select the compounds that are more soluble in acidic solution than in pure water:
Ca(OH)2
Mgą(PO)2
O PbBr,
< Prev
3 of 5
Next >
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 5 images

Recommended textbooks for you


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax



Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
