Decide how the objects in the following table would be listed, if they were listed in order of increasing kinetic energy. That is, select "1" next to the object with he lowest kinetic energy, select "2" next to the object with the second lowest kinetic energy, and so on. object kinetic energy A 6.6 mg insect moving at 7.2 cm/s. (choose one) A 10.0 mg insect moving at 12.4 cm/s. (choose one) A 9.0 mg insect moving at 7.2 cm/s. (choose one) (choose one) A 10.0 mg insect moving at 7.2 cm/s. ? 1 Submit Assignment Continue Pvecy Terms of Use 2019 McGraw-Hill Education. All Rights Reserved. MacBook Pro DII F8 g0 F7 F6 F5 F4 GE X
Decide how the objects in the following table would be listed, if they were listed in order of increasing kinetic energy. That is, select "1" next to the object with he lowest kinetic energy, select "2" next to the object with the second lowest kinetic energy, and so on. object kinetic energy A 6.6 mg insect moving at 7.2 cm/s. (choose one) A 10.0 mg insect moving at 12.4 cm/s. (choose one) A 9.0 mg insect moving at 7.2 cm/s. (choose one) (choose one) A 10.0 mg insect moving at 7.2 cm/s. ? 1 Submit Assignment Continue Pvecy Terms of Use 2019 McGraw-Hill Education. All Rights Reserved. MacBook Pro DII F8 g0 F7 F6 F5 F4 GE X
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter1: Basic Concepts Of Chemistry
Section: Chapter Questions
Problem 14PS
Related questions
Question
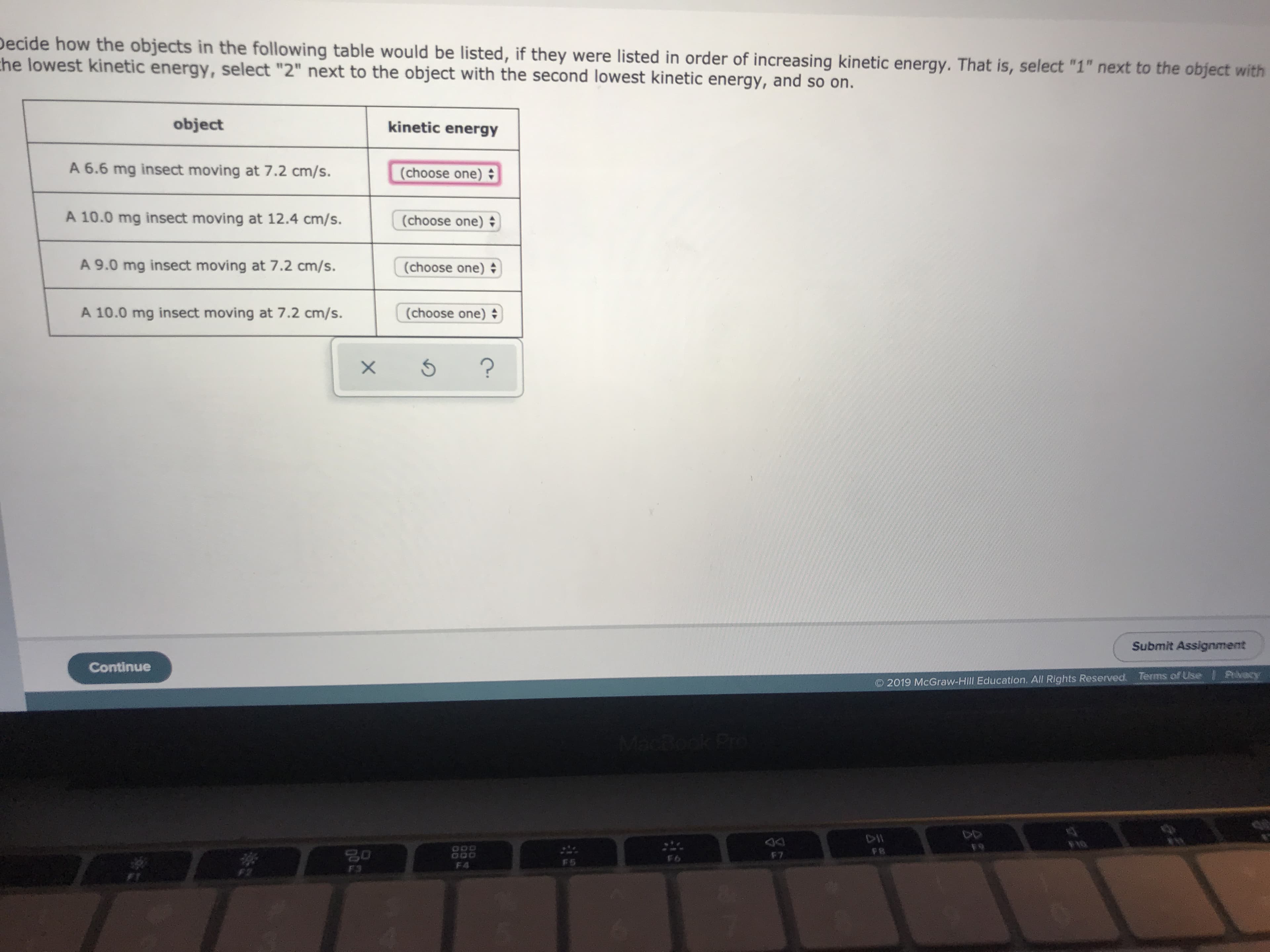
Transcribed Image Text:Decide how the objects in the following table would be listed, if they were listed in order of increasing kinetic energy. That is, select "1" next to the object with
he lowest kinetic energy, select "2" next to the object with the second lowest kinetic energy, and so on.
object
kinetic energy
A 6.6 mg insect moving at 7.2 cm/s.
(choose one)
A 10.0 mg insect moving at 12.4 cm/s.
(choose one)
A 9.0 mg insect moving at 7.2 cm/s.
(choose one)
(choose one)
A 10.0 mg insect moving at 7.2 cm/s.
?
1
Submit Assignment
Continue
Pvecy
Terms of Use
2019 McGraw-Hill Education. All Rights Reserved.
MacBook Pro
DII
F8
g0
F7
F6
F5
F4
GE
X
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 7 steps with 5 images

Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
