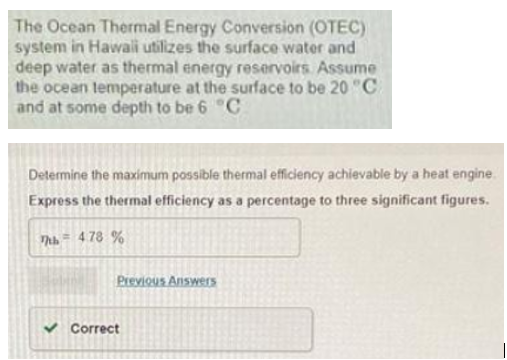
Determine the maximum possible thermal efficiency achievable by a heat engine.
Q: B. A Carnot heat engine, shown in Fig. below, receives 400 kJ of heat per cycle from a…
A: Given:Qa=400 KJTH=552°CTL=25°C
Q: (d) A heat engine operates by taking heat from a thermal reservoir at 1000°C and rejecting heat to a…
A: Given Data: The temperature of the thermal reservoir source is TH = 1000°C The temperature of the…
Q: 6. Calculate the thermal efficiency of a Carnot cycle heat engine operating between reservoirs at…
A: Given data: The temperature of the source, Th = 300°C. The temperature of the sink, Tc = 45°C. The…
Q: A Carnot heat engine receives heat from a reservoir at 900°C at a rate of 800 kJ/min and rejects the…
A:
Q: d) Is that possible? e) Draw a heat engine diagram of a perpetual motion machine. f) Is it possible…
A: following is the answer to the above question
Q: A household refrigerator runs one-fourth of the time and removes heat from the food compartment at…
A: the average rate of heat removal ( ) = 800 kJ/hr (i.e if refrigerator works for one hour then it…
Q: A dealer advertises that he has just received a shipment of electric resistance heaters for…
A: Given data
Q: In a heat engine operating in a cycle between a source temperature of 606°C and a sink temperature…
A: Heat engine is a thermodynamic equipment which converts the thermal heat energy into useful work.…
Q: A heat engine is operating on a Carnot cycle and has a thermal efficiency of 55 percent. The waste…
A: To find out the power output Heat rejected = 800 BTU/min…
Q: ) A heat pump is used to heat a house and maintain it at 25°C. On a winter day when the outdoor air…
A:
Q: A heat engine operates between a source at 477°C and a sink at 25°C. If heat is supplied to the heat…
A:
Q: Consider two Carnot heat engines operating in series. The first engine receives heat from the…
A:
Q: A thermodynamicist claims to have developed a heat pump with a COP of 1.7 when operating with…
A: Given data as per question Lower temperature = 273 k Higher temperature 293 k
Q: A carnot heat engine with thermal efficiency of 70% is supplied with heat at a rate of 800 kJ/min.…
A:
Q: Two Carnot heat engines are operating in series such that the heat sink of the first engine serves…
A: Given: The source temperature of the first engine is T1=1300 K. The sink temperature of the second…
Q: A heat pump is to be used for heating a house in winter. The house is to be maintained at 78°F at…
A: Given: The temperature of the house is T1=78 °F The temperature of the outdoor is T2=25 °F The heat…
Q: A heat pump is to be used for heating a house in winter. The house is to be maintained at 78°F at…
A: Given, T1 = 78°F T2 = 50°F Tambient = 25°F Q = 70,000 Btu/h COP = 11-(T2T1)COP =…
Q: A
A:
Q: Heat is transferred to a heat engine from a furnace at a rate of 80 MW. If the rate of waste heat…
A: Given, Heat from furnace (QH) = 80 MW Heat loss (QL )= 50 MW
Q: Q1// A heat engine operates between a source at 550°C and a sink at 25°C. If heat is supplied to the…
A:
Q: In an effort to conserve energy in a heat-engine cycle, somebody suggests incorporating a…
A: Given: In order to conserve energy in a heat engine cycle, incorporating a refrigerator that will…
Q: Consider two Carnot heat engines operating in series. The first engine receives heat from the…
A: Given Data T1=1400 K T3=300 K
Q: A Carnot device operates between a cold reservoir of temperature 173 K and a hot reservoir of…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: A Carnot heat engine operates between a source at 727 °C and a sink at 27 °C If the heat engine is…
A:
Q: What is the Carnot efficiency of an automobile engine which supplies heat at 800 K and reject heat…
A: Carnot cycle is an imaginary cycle that gives the maximum possible efficiency between two…
Q: What is cyclic Heat engine? What is the difference between refrigerator and heat Pump?
A:
Q: A Carnot heat engine receives heat from a reservoir at 900°C at a rate of 800 kJ/min and rejects the…
A: Given: The temperature of the reservoir is T1=900°C. The temperature of the refrigerated space is…
Q: Explain the principle of refrigerator? Show that a reversible heat engine acts as a refrigerator.
A: Principle of refrigeration: Refrigeration is the cooling of a system below the temperature of its…
Q: A heat engine having an efficiency of 70% is used to drive a refrigerator having a coefficient of…
A:
Q: What is the highest possible theoretical efficiency of a heat engine operating with a hot reservoir…
A: Given:- T1=2000°C T2=10°C To find:- The theoretical efficiency of heat engine
Q: B. A Carnot heat engine, shown in Fig. below, receives 400 kJ of heat per cycle from a…
A: Given question belongs to the subject Engineering Thermodynamics from the topic Second Law of…
Q: A Carnot heat engine operates between a source at 1000 K and a sink at 300 K. If the heat engine is…
A:
Q: A heat engine with a thermal efficiency of 45 percent rejects 1500 kJ / kg of heat. How much heat…
A:
Q: The efficiency of a heat engine is working between temperature T1 and T2 (T1 > T2) is
A: Given:- Heat engine Higher temperature=T1 Lower temperature=T2 T1>T2 To find:- Efficiency
Q: A heat engine with a thermal efficiency of 40 percent rejects 1000 kJ/kg of heat. Determine the…
A:
Q: Your colleague from a non-thermodynamics background, has asked you to explain to him/her how the…
A: The first law of thermodynamics deals with the quantitative conversion of heat into work. Whereas…
Q: What is the difference between refrigerators and heat pumps? From their mechanics and theory
A: The heat pump and refrigerator can be described as: Refrigerator: The refrigerator is a device that…
Q: Two (2) Carnot engines are arranged in the manner that the latter engine receives the heat rejected…
A:
Q: Illustrate the contradiction between refrigeration cycles and the second law of thermodynamics while…
A:
Q: Two (2) Carnot engines are arranged in the manner that the latter engine receives the heat rejected…
A:
Q: Determine the minimum work per unit of heat transfer from the source reservoir that is required to…
A: To find The minimum work required to drive a heat pump W. Given The temperature of the sinkTh is…
Q: An innovative way of power generation involves the utilization of geothermal energy—the energy of…
A:
Q: A heat engine operates between two reservoirs at 800 and 20°C. One-third of the work output of the…
A:
Q: A heat engine produces 50 kW of power while consuming 50 kW of heat from a source at 1390 K, 70 kW…
A:
Q: A heat engine has a total heat input of 1.3 kJ and a thermal efficiency of 35 percent. How much work…
A:
Q: What is the highest possible theoretical efficiency of a heat engine operating with a hot reservoir…
A: Given Data: The temperature of hot reservoir furnace is, T1=2000°C=2000+273 K=2273 K. The…
Learn your way
Includes step-by-step video
Step by step
Solved in 2 steps

- The heat transfer coefficient between a surface and a liquid is 57 W/(m2K). How many watts per square meter will be transferred in this system if the temperature difference is 10C?Heat is transferred at a rate of 0.1 kW through glass wool insulation (density=100kg/m3) with a 5-cm thickness and 2-m2 area. If the hot surface is at 70C, determine the temperature of the cooler surface.The condenser of a steam power plant operates at a pressure of 7.38 kPa (Tsat = 40°C). Steam at this pressure condenses on the outer surfaces of horizontal pipes through which cooling water circulates. The outer diameter of the pipes is 3 cm, and the outer surfaces of the pipes are maintained at 30°C. Determine the rate of heat transfer to the cooling water circulating in the pipes.The properties of water at the saturation temperature of 40°C are: hfg = 2407 × 10^3J/kg and ρv=0.05 kg/m3. The properties of liquid water at the film temperature: ρl = 994 kg/m3, Cpl =4178 J/kg°C, µl = 0.72 × 10^-3 kg/m.s, kl = 0.623 W/m°C Select one: a. 6758W b. 5758W c. 8758W d. 7758W
- The net radiant-heat exchange between a 20-by-20 ft rough plaster panel-heated ceiling and a 20-by-10-ft side wall, which has an emissivity of 0.80, is 2400 Btu/hr. If the surface temperature of the ceiling is 120 deg. F, determine the average surface temperature of the side wall.What are the three methods of Heat Transfer? Explain each.65 kg/min of water is heated from 20°C to 80°C passing through the duct of 3 cm x 2 cm. The duct is heated by condensing the steam on its outer surface. Find out the length of the duct required. Properties of water, p = 995 kg/m³, µ = 7.65 × 10-kg/m-s C = 4.174 kJ/kg-K, K = 0.623 W/m-K Conductivity of duct material = 35 W/m-K Use the following correlations: Nu = 0.023 Re0.8 Pr0.4 for turbulent flow nu 4.36 for laminar flow
- Q#01: Write a short note on the following: What are the best possible solutions to increase the heat transfer rate of a body? Which material will transfer more heat? Either a material with thermal conductivity ? = 121 or ? = 22 . Justify your answer.A coal with a higher heating value of 6,700 Kcal/kg is consumed at a rate of 600 kg/hr. in a water tube steam generator with a rated boiler horsepower of 200 HP. The feedwater temperature is 82°C and steam generated is at 11 kg/cm2 absolute, saturated. The developed boiler horsepower is equivalent to 305 HP. Determine: a. Heating surface area in m²b. Steam evaporated in kg/hrc. ASME evaporating units in J/hrd. Percent ratinge. Factor of evaporationf. Over-all thermal efficiencyg. actual specific evaporationh. Equivalent specific evaporation. An automobile engine consumes fuel at a rate of 22 L/h and delivers 55 kW of power to the wheels. If the fuel has a heating value of 44,000 kJ/kg and a density of 0.8 g/cm3, determine the efficiency of this engine. Hint, Qa= mass of fuel * heating value. Draw and label the P-V and T-S diagrams.
- An automobile engine consumes fuel at a rate of 22 L/h and delivers 55 kW of power to thewheels. If the fuel has a heating value of 44,000 kJ/kg and a density of 0.8 g/cm3, determinethe efficiency of this engine. Hint, Qa= mass of fuel * heating value. Answer: 25.57%An automobile engine consumes fuel at a rate of 22 L/h and delivers 55 kW of power to thewheels. If the fuel has a heating value of 44,000 kJ/kg and a density of 0.8 g/cm3, determinethe efficiency of this engine. Hint, Qa= mass of fuel * heating value.An automobile engine consumes fuel at a rate of 22 L/h and delivers 55 kW of power to the wheels. If the fuel has a heating value of 44,000 kJ/kg and a density of 0.8 g/cm3, determine the efficiency of this engine. Hint, Qa= mass of fuel * heating value. And draw the P-V and T-S diagram.

