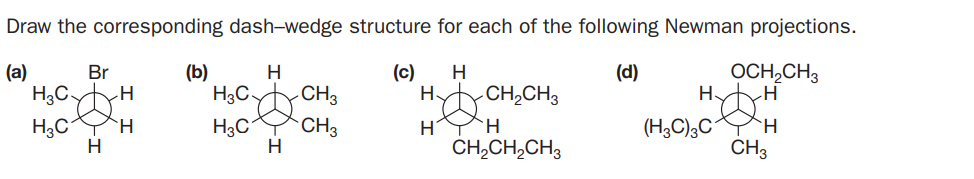
Draw the corresponding dash-wedge structure for each of the following Newman projections. (a) OCH,CH3 H. Br (b) H (c) H (d) HgC HyCH,CH3 H3C H H3C HYH CH,CH,CH3 CH3 (H3C),C CH3 H. H
Q: most taci cofommer below CH3 CH3 OCHS OCHS OC,Hs CH3 OCHS CH3 CH3 OCHS OCHS CH, OCeHs H3C- CH3 CH3…
A: In the given cyclohexane derivative, the presence of substituents and their size decides the…
Q: Consider the following structures: A. Н. H₂C CH₂ H H B. H.CCH, H₂CH H H CH₂ CHS CH₂ D. H₂C HCH₂ CH,…
A: To solve this problem, we need to know the eclipsing and gauche interaction energy due to CH3 and H.
Q: NH2 Br "COOH NC ICH3 ОН H3C H A H HOH,C OCH3 СООН D
A: In a given question , we have to find out absolute configuration which is R/S nomenclature. For R/S…
Q: 2. i) Arrange the following Newman projections of butane in order of their energy level ii) identify…
A:
Q: 3. Assign each molecule or ion to its proper point group. a. 1,2-dibromoethane (in the gauche…
A: For determining the point group, one has to first draw the structure of the compound followed by…
Q: Which of the Newman projections below would correspond to the following molecule from the…
A: In this question we have to tell the Newman projection of the given molecule.
Q: The Newman projection looking down the indicated bond in the following species is best represented…
A: The given species contains two chiral carbon and the structure of the species is as follows:
Q: For 1,2-dichloroethane: a.Draw Newman projections for all eclipsed conformations formed by rotation…
A: 1,2-dichloroethane is represented as:
Q: Which of the following Newman projections correctly represents the structure of (2R,35)-2-chloro-3-…
A: Newman projection visualizes the conformation of a chemical bond from front to back, with the back…
Q: which one has the most red-shifted C-O NH (a) F (b) D K G. H. (d) (c)
A:
Q: Molecular Formula: C8H03 Compute for the DU (degree of unsaturation) Then draw the structure of the…
A:
Q: 6. For the following compound, (1) draw the complete Lewis structure; (2) site down the C3- C4 bond…
A: The given compound is: (1) Draw the complete Lewis structure. (2) Site down the C3-C4 bond axis and…
Q: Draw the Newman pr0jecti0ns 0f the three p0ssible staggered c0nf0rmati0ns 0f 2,3-dimethylbutane,…
A: Newman projection is useful in alkane steriochemistry visualizes the conformation of a chemical bond…
Q: Draw the possible constitutional isomers having molecular formula C8H16 that meet all of the…
A: In order to know constitution isomers we need to determine the degrees of unsaturation:number of pi…
Q: (a) (b) (c) H₂CCH3 CH3 I CAH CH3 CH3 or or or H₂C CH₂CH3 CH3 H3C CH3 CH3
A: The equitorial substitutents are more stable due to less steric repulsions .
Q: For 1-bromopropane, which of these staggered Newman projections has the lowest energy? CH3 Br Br. Br…
A:
Q: A Newman projection of a disubstituted cyclohexane is shown below. etermine which of the following…
A:
Q: bond-line structure is represented by the Newman projection below
A:
Q: 6. Provide the complete mechanism (arrow, lone pairs, formal charges) for the following…
A:
Q: Nhen compound X undergoes ring-flipping, which compound below represents its ring-flipped conformer?…
A: We have given the organic compound i.e. 1,2diethyl-3-methyl cyclohexane ring we have to gives its…
Q: Which of the Newman projections below would correspond to the following molecule from the…
A:
Q: Which molecules have (Z)- ? configuration H3C CH2CH Br, HO;C H CI CI CH3 Br H3C CH;CH, H3C CH2CH3 H'…
A: In E configuration, two bulkier groups in same side of plane.
Q: Which of the following structure represents the three dimensional line structure of the Newman…
A:
Q: Which of the following electrostatic potential maps best represents nitromethane (CH3NO2)? Explain.…
A: A covalent bonding will turn into a polar covalent bonding only if there exists a highly…
Q: Which of the following molecules is (are) E configuration? HOH2C CH3 HOH2C у. NC CH3 OMe Z. х. H3C…
A: Given molecules,
Q: (b) Identify structure(s) for all constitutional isomers with the molecular formula C4H3 that have…
A: Isomers having same molecular formula but different connectivity are called constitutional isomers.…
Q: The central carbon atom of an allene is a member of two double bonds, and ithas an interesting…
A: An orbital diagram of allene and the two perpendicular ends are shown below.
Q: b. of butane-2,3-diamine of which the semi-developed formula is: NH2 13C-CH-CH-CH3 NH2 in…
A: In hashed-wedged projection, there is the representation of bonds in the form of a backline called a…
Q: Consider the molecule whose line structure is below: Long Long wochain www chain The molecule under…
A:
Q: Rank the following sets of substituents according to the Cahn–Ingold, Prelog sequence rules: (а)…
A: Since you have asked multiparts questions, we will solve the first three sub parts questions for…
Q: 4 CI IS - Consider the following structure. Convert the Newman projection to a bond-line formula…
A: -> In bond line formula there are not seen any carbon and hydrogen in the structure . -> For…
Q: Which is/are correct? a.The dipole moment of propanal is greater than that of 1-butene. b.Carbonyl…
A: There are some statements.... a) The dipole moment of propanal is greater than that of 1-butene. b)…
Q: The most stable conformation of line structure X is HO ОН X iPr Me H. iPr iPr Me H. 'Pr Me. iPr H…
A:
Q: 1. What is the difference between conformational rotation and symmetry rotation? Consider Newman…
A: Conformational rotation :- The rotation of molecule about a single bond which results in the…
Q: Is the following compound R or S. Support your claim by indicating the priority ofthe groups…
A: R,S-nomenclature of the compounds is done according to the cahn-ingold prelog rules according to…
Q: Answer the following questions for 1,3,5- hexatriene, the conjugated triene containing six…
A: 1,3,5- hexatriene has the conjugated triene containing six carbons structure.
Q: Answer the following questions for 1,3,5- hexatriene, the conjugated triene containing six…
A: The hexatriene has six carbon atoms with alternating double bonds. In a molecule, the atomic and…
Q: CH3 а. (H3C)3C- SF3 H3C- CO2H CH b. H3C- H3C CO2H N-SF2 BF, H3C- (H3C)2HC ,Ni(OH);; К,СО,, DMSO -NH2…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. If you need help with other sub…
Q: Which of the following Newman projections correctly represents the structure of (2R,3S)-2-chloro-…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Arrange the following groups in order of decreasing priority according to R-S…
A: The rules that must be followed while prioritizing the substituent group for R-S configurations are-…
Q: Do the following molecule contain conjugated double bonds?
A: A conjugated double bond is defined as that bond which are in conjugation to each other and hence…
Q: Which of the following Newman projections represents (2R,3R)-dibromobutane?
A:
Q: 3. Convert the following perspective drawing into Newman projections by looking along the bond as…
A: Note : Put dashed bonds left side on Newmann projection. Put wedge bonds right side on Newmann…
Q: 8. What type of orbitals, from each double carbon, overlan to from the o-bond within the double?…
A: Answer
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 5 images

- Consider the Newman projection below. a. Draw a full Lewis structure of this molecule with R1=Me,R2=Et , and R3=iPr . b. Given the sizes of these R groups (R3R2R1) , does the Newman projection above show thelowest potential energy conformation of this bond? If not, draw a Newman projectionshowing the lowest P.E. conformation (sighting down this same bond). c. To draw a Newman projection in the lowest P.E. conformation, the following rule of thumbusually applies: Place the largest group on the front carbon anti to the largest group on theback carbon. Is your answer to the previous question consistent with this rule of thumb?translate the bond-line notation structure to the Newman projection by filling int the missing groups (A, B, C, D or E) on the lines in the Newman projection so they match the conformation given in the original structure. Circle if the conformation is a staggered or eclipsed.For the compounds given (A, B, C) 1. determine their relationship ( enantimers, idential/same compind, diatermeros, constitutional isomers) and give a short explanation why
- the stereo center in this molecule is ___. a. R b. SAnd can u show with the arrow formalism? And can u pls tell me what the role of Hcl and H2O is.? Thank u.....Draw the Newman pr0jecti0ns 0f the three p0ssible staggered c0nf0rmati0ns 0f2,3-dimethylbutane, viewed thr0ugh the C2—C3 b0nd. What are the relativeenergies of each c0nf0rmati0n?
- Which of the attached is (are) possible Newman projections for 2-methylpentane?Is the 1st compound ( more, less or equally) stable than B? Pls show through illustrations of chair conformationsAnswer the following question for 1,3,5- hexatriene, the conjugated triene containing six carbons. Which MOs are the frontier molecular orbitals?
- Then draw the most stable and least stable Newman projection conformation from the C4-C5 bond in the molecule aboveHow many carbon atoms in this remdesivir are considered chiral? Please circle each one.Answer the following questions for 1,3,5- hexatriene, the conjugated triene containing six carbons.Which MOs are bonding? Which are antibonding?

