Q: Topic: Isolation of Crude Ovalbumin from Egg White by Ammonium Sulfate Precipitation (Salting Out)…
A: The experiment is about isolation of crude ovalbumin, which is a protein.
Q: Q: 1: The distribution constant for iodine between an organic solvent and HO is 85. Find the…
A: From given The distribution constant for iodine between an organic solvent and H2O is 85. We need…
Q: What Is The Activity Coefficient Of Benzoate Ion (C7H5O2-) With An Ionic Size Parameter Of a = 0.60…
A:
Q: 1. Will a 5% preparation of caffeine citrate compounded in citrate buffer, pH 4.8 with no additional…
A:
Q: In the ternary phase diagram experiment acetic acid (7.6 mL) and water (1.4 mL) mixture is titrated…
A:
Q: What is the purpose of acetone in the final filtration of the precipitate in Gravimetric…
A: The purpose of acetone in the final filteration of the precipitate in gravimetric determination of…
Q: 4- If Ksp of Agl = 8.3x10-17 calculate the solubility of Agl by gm/L ? 234. 8 x9. 11x10-6 gm/L O2.34…
A:
Q: The Kjedahl procedure was used to analyze 256 µL of a solution containing 37.9 mg protein/mL. The…
A: NH3 + HCl = NH4Cl HCl + NaOH = H2O + NaCl Here, one mole of NH3 reacts with one of HCl and also…
Q: In the lab, Annie mixed 2.5 g of the benzoic acid with 10.00mL of 1.000M NaOH and diluted to 100mL.…
A: The reaction of an acid with a base will result in the formation of salt and water. For example, the…
Q: Explain how preconcentration of cations with an ion exchanger works. Why must the concentrated acid…
A: Ion exchange chromatography involves the reversible exchange of ions between a solid phase (that is…
Q: Zinc uranyl acetate, Magnesium uranyl acetate and Cobalt uranyl acetate, forms insoluble salts with…
A: zinc uranyl acetate, magnesium uranyl acetate, and cobalt uranyl acetate, forms insoluble salt with…
Q: Write the balancing equations for C,H, and O.:)
A: Since you have asked multiple questions, we will solve first one for you. For remaining question…
Q: Benzoic acid is not very soluble in water and has a much stronger affinity for organic solvents.…
A: Organic compounds have a greater tendency to soluble in organic solvents and sodium salts have a…
Q: Determine concentration of OH- in a 0.724 M solution of Bro- (Kb = 4.0 × 10-6). PREV 2 Based on your…
A:
Q: What is the minimum distribution constant that permits removal of 99% of a solute from 50.0 mL of…
A: What is the minimum distribution constant that permits removal of 99% of a solute from 50.0 mL of…
Q: 1. What type of precipitate is BaSO4? 2. Why is a filter paper preferred over a sintered glass…
A: 1) Barium sulphate is a white color precipitate , it is ionic in nature . when barium chloride is…
Q: . After the reflux period, what substances are pre n Whu will the density of the organic phase be…
A: In organic chemistry, Reactants are mixed in a solvent media, and products are obtained. The final…
Q: What weight of strontium acetate containing 99% Sr(CH3COOH)2.½H2O must be used as sample in an assay…
A: Solution -
Q: Consider the reaction below. If the solution mixture contains 0.15M HC 2H 30 2 what is the…
A: Equilibrium constant is the ratio of molar concentration of products to the molar concentration of…
Q: Calculate the percent error in Avogadro's number calculated by using the following experimental…
A: Concentration of stearic acid = 1.2×10-4gram/milliliter.Number of drops to complete monolayer = 23…
Q: . A 1.2-gram sample of lanolin was treated with Wij’s solution and excess potassium iodide solution.…
A: GIVEN- Mass of lanolin sample(W)=1.2g Normality of sodium thiosulfate solution=0.1N Volume of…
Q: If K-2.35 & K,- 1.32 x 1010 Suppose 50ml of 0.010M aqueous amine is extracted with 100ml of benzene.…
A: Given: K = 2.35 Ka=1.32×10-10 Volume of aqueous amine = 50 mL Volume of benzene = 100 mL…
Q: Using Equation 5.9, show that three extractions with 5-mL portions of a solvent give better recovery…
A:
Q: What is the minimum distribution constant that permits removal of 99% of a solute from 50.0 mL of…
A: Distribution law gives information about the partition of a solute between two immiscible phases as…
Q: The Kjedahl procedure was used to analyze 256 µL of a solution containing 37.0 mg protein/mL. The…
A: Given that,Volume of HCl = 5.00 mL.Molarity of HCl = 0.0336 M.Volume of NaOH = 6.34 mL.Molarity of…
Q: FeCl3 and MgCl2 solution was prepared by dissolving FeCl3 (AR) and MgCl2 (AR) in distilled water.…
A: Hydrolysis is a process in which the compound reacts with water. The required compound in this given…
Q: For separating the 9H-fluorene and benzophenone mixture, you used silica gel as your solid phase,…
A: Column chromatography is an analytical tool that is employed in the separation of the mixture of…
Q: What is the minimum distribution constant that permits removal of 99% of a solute from 50.0 mL of…
A: Solution - According to the question - Given - It is given that 99% of given solute is removed from…
Q: Accurately prepare a 50cm stock solution of (approx.) 0.003M potassium permanganate solutic Very…
A: Given that: the volume of potassium permanganate solution = 50 cm3 the volume of potassium…
Q: Why is an aqueous solution of NaCl used to prepare the solutions for runs 2-4 instead of deionized…
A: Deionized water contains no ions in it where as the sodium chloride solution having sodium(Na+) ion…
Q: Prepare 25ppm of Cd (NO3 )2.4H2O in a 500ml from 1000ppm stock solution of Cd (NO3)2.4H2O
A:
Q: Order the compound A, B and C from lowest to highest Rr. The sample is spotted on a silica te and…
A: Retardation factor (Rf) is calculated by the distance travelled by solute divided by distance…
Q: Question 3
A: The total mass of the mixture (Msol) is calculated as shown in equation (1) where VX and VY are the…
Q: Why should deionized water he used throughout the analysis?
A: Metal ions are formed by loss of electrons from metal. Electrons are negatively charged particles…
Q: The quantitative determination of pyridoxine hydrochloride can be carried out by methods: A.…
A: A multiple choice question based on coordination compounds that is to be accomplished.
Q: Ice with 2 N HCl and 5% NaNO2 of aromatic primary - aliphatic secondary and tertiary amines write…
A:
Q: Quetion 2 The solubality preduct of Cu(OH), i5 4.8 x 10-20. Calculate the value of pCu", or leg[Cu…
A: Common ion effect: The common ion effect refers to the decrease in solubility of an ionic ppt by the…
Q: Given a solution containing 0.010 M CaCl2 and 0.020 M MgCl2, what formula should be used to…
A: Concept: Debye huckle limiting law: Debye-Hückel developed a model for calculating activity…
Q: If Vsu= Vorg, how many times should the extraction process be repeated so that 1 g of substance with…
A: Let the required number of extractions be 'n'.
Q: Why must the solution be saturated when its solubility is determined?
A: Introduction: Solubility product: Solubility product constant measure the extent to which a solute…
Q: NaCl equivalent of a 1% solution of pilocarpine nitrate
A: In this question we have to calculate NaCl equivalent of 1% solution of pilocarpine nitrate. Also we…
Q: The standardization of same titrant during the determination of BOD5 of water sample was done…
A: The BOD5 test, also known as the Biochemical Oxygen Demand test, is a biochemical test that measures…
Q: 17. 1-Octanol is immiscible with water. Consider the octanol/water distribution constants for a…
A: The question is based on the concept of solvent extraction. we have to identify the correct…
Q: 1.What is the significance when KD is either a small or large value? 2. Methyl alcohol and ethyl…
A: Hello. Multiple questions have been posted. The first question has been solved. To receive solutions…
Q: One of the components of nutmeg is an organic compound, Trimiristin, A. To extract this compound…
A: One of the components of nutmeg is an organic compound, Trimiristin, A. To extract this compound…
Q: Write all the steps of recrystallization of impure naphthalene from methanol. How to identify the…
A: In this question, we have to find out the correct answer of given problem by the help of…
Q: In a reaction involving the iodination of acetone, the following volumes were used to make up the…
A: Here the mixture contains 5ml of 3.40M acetone, 10 ml of 1.50M HCl ,10 ml of 0.004M and 25 ml water.…
Q: On the direct iodometric titration experiment, Standardization of Na2S2O3 with KIO3 standard…
A: One of the most effective redox titration methods is iodometry. Because of its relatively low pH,…
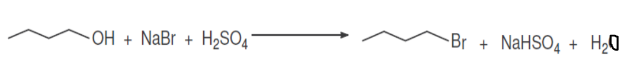
During the formation of the n-butyl bromide, a washing is carried out with the NaHCO3 solution and 2 phases are formed again. What is the organic phase? Justify
Step by step
Solved in 2 steps with 1 images


