Ethyl alcohol, CH;CH,OH, was one of the first organic chemicals deliberately synthesized by humans. It has many uses in industry, and it is the alcohol contained in alcoholic beverages. It can be obtained by the fermentation of sugar or synthesized by the hydration of ethylene in the following reaction: H H H H C=C + H-0: Н—с—с—о—н H H Using the bond energies in Table 7.3, calculate an approximate enthalpy change, AH, for this reaction. Average Bond Lengths and Bond Energies for Some Common Bonds Bond Bond Length (A) Bond Energy (kJ/mol) C-C 1.54 345 C= C 1.34 611 C= C 1.20 837 C-N 1.43 290 C= N 1.38 615 C= N 1.16 891 C-O 1.43 350 C= 0 1.23 741 C= 0 1.13 1080 Table 7.3
Ethyl alcohol, CH;CH,OH, was one of the first organic chemicals deliberately synthesized by humans. It has many uses in industry, and it is the alcohol contained in alcoholic beverages. It can be obtained by the fermentation of sugar or synthesized by the hydration of ethylene in the following reaction: H H H H C=C + H-0: Н—с—с—о—н H H Using the bond energies in Table 7.3, calculate an approximate enthalpy change, AH, for this reaction. Average Bond Lengths and Bond Energies for Some Common Bonds Bond Bond Length (A) Bond Energy (kJ/mol) C-C 1.54 345 C= C 1.34 611 C= C 1.20 837 C-N 1.43 290 C= N 1.38 615 C= N 1.16 891 C-O 1.43 350 C= 0 1.23 741 C= 0 1.13 1080 Table 7.3
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter8: Bonding And Molecular Structure
Section: Chapter Questions
Problem 53PS: Hydrogenation reactions, which involve the addition of H2 to a molecule, are widely used in industry...
Related questions
Question
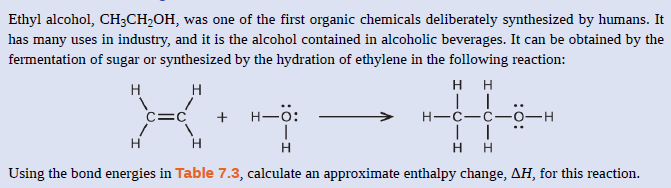
Transcribed Image Text:Ethyl alcohol, CH;CH,OH, was one of the first organic chemicals deliberately synthesized by humans. It
has many uses in industry, and it is the alcohol contained in alcoholic beverages. It can be obtained by the
fermentation of sugar or synthesized by the hydration of ethylene in the following reaction:
H H
H
H
C=C
+ H-0:
Н—с—с—о—н
H
H
Using the bond energies in Table 7.3, calculate an approximate enthalpy change, AH, for this reaction.
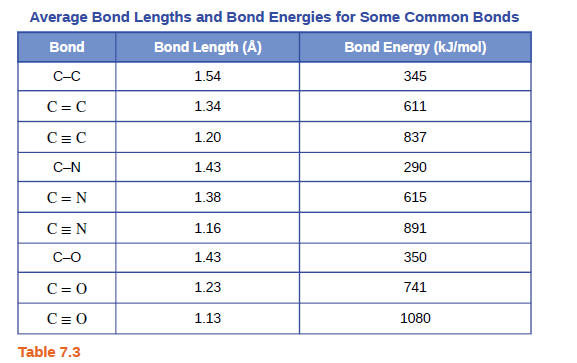
Transcribed Image Text:Average Bond Lengths and Bond Energies for Some Common Bonds
Bond
Bond Length (A)
Bond Energy (kJ/mol)
C-C
1.54
345
C= C
1.34
611
C= C
1.20
837
C-N
1.43
290
C= N
1.38
615
C= N
1.16
891
C-O
1.43
350
C= 0
1.23
741
C= 0
1.13
1080
Table 7.3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 1 images

Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning