Experiment No. 13 GRAVIMETRIC DETERMINATION OF AN UNKNOWN SULFATE Table 1. Experimental Data Mass of empty 250 ml beaker 98.5342 g Mass of 250-ml beaker and unknown 98.9070 g sulfate Mass of unknown sulfate Mass of empty crucible with cover and 51.6667 g barium sulfate Mass of crucible with cover and barium 52.4802 g sulfate Mass of barium sulfate . Calculate the mass of sulfate in the unknown sample. Show your calculations. . Calculate the percentage by mass of sulfate in the unknown sample. Show your
Experiment No. 13 GRAVIMETRIC DETERMINATION OF AN UNKNOWN SULFATE Table 1. Experimental Data Mass of empty 250 ml beaker 98.5342 g Mass of 250-ml beaker and unknown 98.9070 g sulfate Mass of unknown sulfate Mass of empty crucible with cover and 51.6667 g barium sulfate Mass of crucible with cover and barium 52.4802 g sulfate Mass of barium sulfate . Calculate the mass of sulfate in the unknown sample. Show your calculations. . Calculate the percentage by mass of sulfate in the unknown sample. Show your
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 7P
Related questions
Question
100%
Complete the table and answer the following questions.
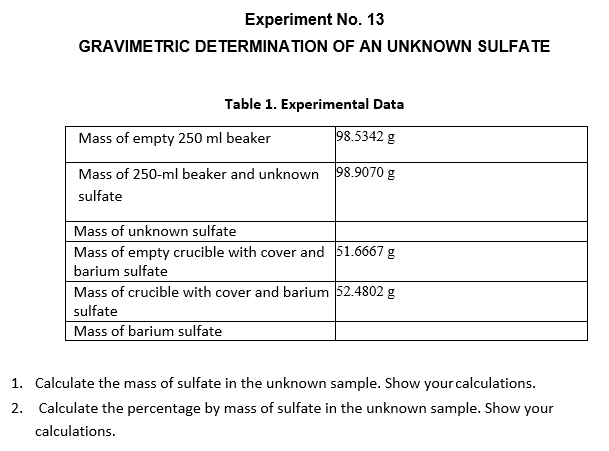
Transcribed Image Text:Experiment No. 13
GRAVIMETRIC DETERMINATION OF AN UNKNOWN SULFATE
Table 1. Experimental Data
Mass of empty 250 ml beaker
98.5342 g
Mass of 250-ml beaker and unknown 98.9070 g
sulfate
Mass of unknown sulfate
Mass of empty crucible with cover and 51.6667 g
barium sulfate
Mass of crucible with cover and barium 52.4802 g
sulfate
Mass of barium sulfate
1. Calculate the mass of sulfate in the unknown sample. Show your calculations.
2. Calculate the percentage by mass of sulfate in the unknown sample. Show your
calculations.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

