* Explain exactly what experiment(s) must be performed in order to evaluate the activation energy (E ) for this reaction. What data must be plotted? 00 0EGO00 Name Time M T W R F The data given in the table below is for the following reaction (run at 25°C): 2 NO + O,l) → 2 NO 2(g) (), 2(g) Initial Concentration (M) Initial rate Experiment Mol/(L s) NO 02 0.0010 0.0010 7 X 10-6 0.0010 0.0020 14 X 106 3 0.0010 0.0030 21 X 106 0.0020 0.0030 84 X 106 0.0030 0.0030 189×10-6
* Explain exactly what experiment(s) must be performed in order to evaluate the activation energy (E ) for this reaction. What data must be plotted? 00 0EGO00 Name Time M T W R F The data given in the table below is for the following reaction (run at 25°C): 2 NO + O,l) → 2 NO 2(g) (), 2(g) Initial Concentration (M) Initial rate Experiment Mol/(L s) NO 02 0.0010 0.0010 7 X 10-6 0.0010 0.0020 14 X 106 3 0.0010 0.0030 21 X 106 0.0020 0.0030 84 X 106 0.0030 0.0030 189×10-6
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter12: Kinetics
Section: Chapter Questions
Problem 82E: For each of the following reaction diagrams, estimate the activation energy (Ea) of the reaction:
Related questions
Question

Transcribed Image Text:* Explain exactly what experiment(s) must be performed in order to evaluate the activation
energy (E ) for this reaction. What data must be plotted?
00
0EGO00
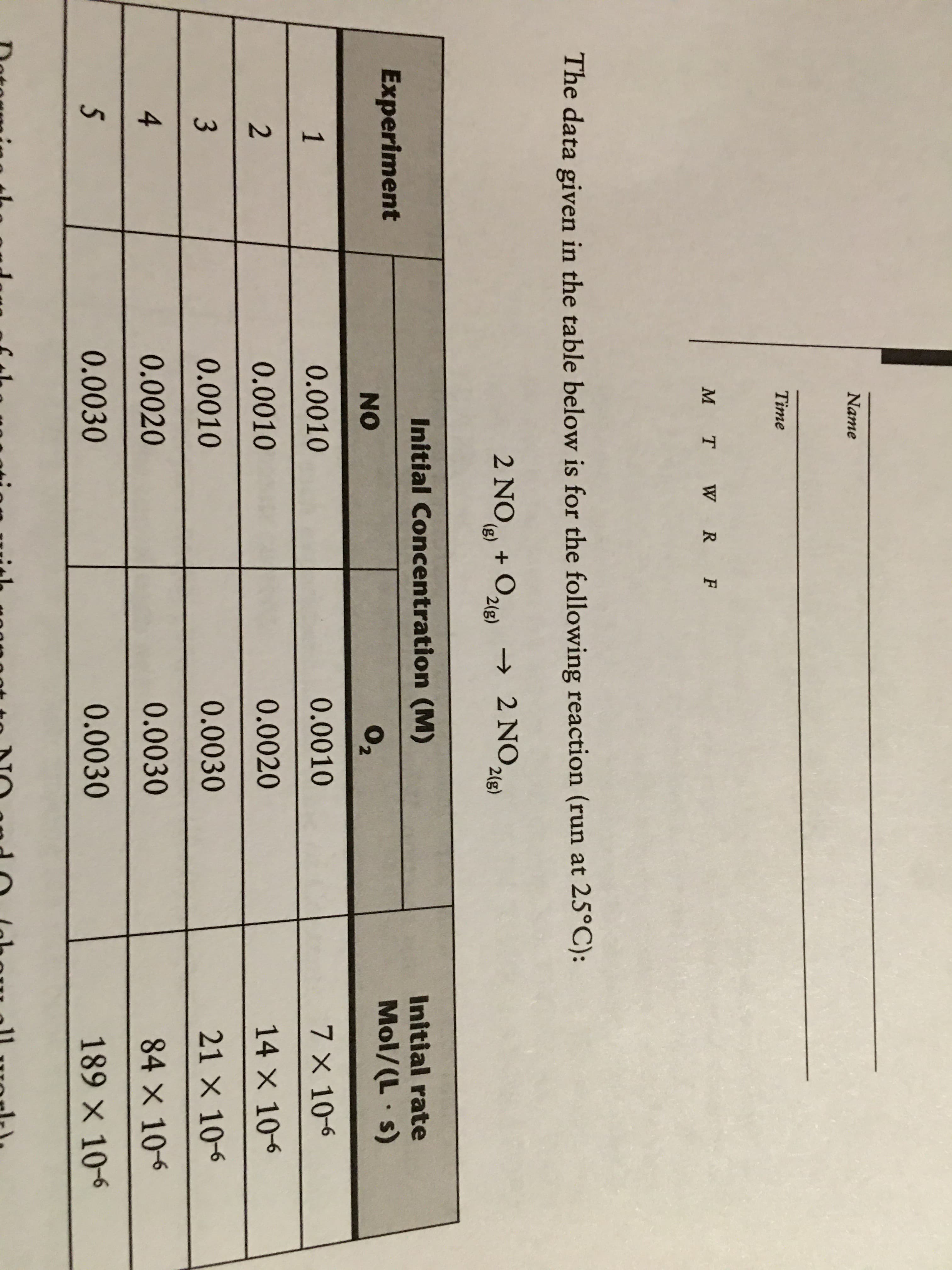
Transcribed Image Text:Name
Time
M T W R F
The data given in the table below is for the following reaction (run at 25°C):
2 NO + O,l) → 2 NO
2(g)
(),
2(g)
Initial Concentration (M)
Initial rate
Experiment
Mol/(L s)
NO
02
0.0010
0.0010
7 X 10-6
0.0010
0.0020
14 X 106
3
0.0010
0.0030
21 X 106
0.0020
0.0030
84 X 106
0.0030
0.0030
189×10-6
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning
