Figure 3-9. 100 D 4000 100 50 (715) 100 4000 50 4900 m www A 3000 3000 3000 B 2000 2008 C 2000 SAVEHUN MYEHURI- NAVENUER 1100 1500 1900 1000 1000 1000 100 500
Figure 3-9. 100 D 4000 100 50 (715) 100 4000 50 4900 m www A 3000 3000 3000 B 2000 2008 C 2000 SAVEHUN MYEHURI- NAVENUER 1100 1500 1900 1000 1000 1000 100 500
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.9QAP
Related questions
Question
Transcribed Image Text:Figure 3-9.
100
FUNSHINE
4800
001
Linti
4000
100
4900
3000
3000
3000
B
2008
2008
C
2008
HAVENUMER
NAVENURIER-)
NAVENUTEERIT
CUST
1300
1900
1000
1000
1000
500
500
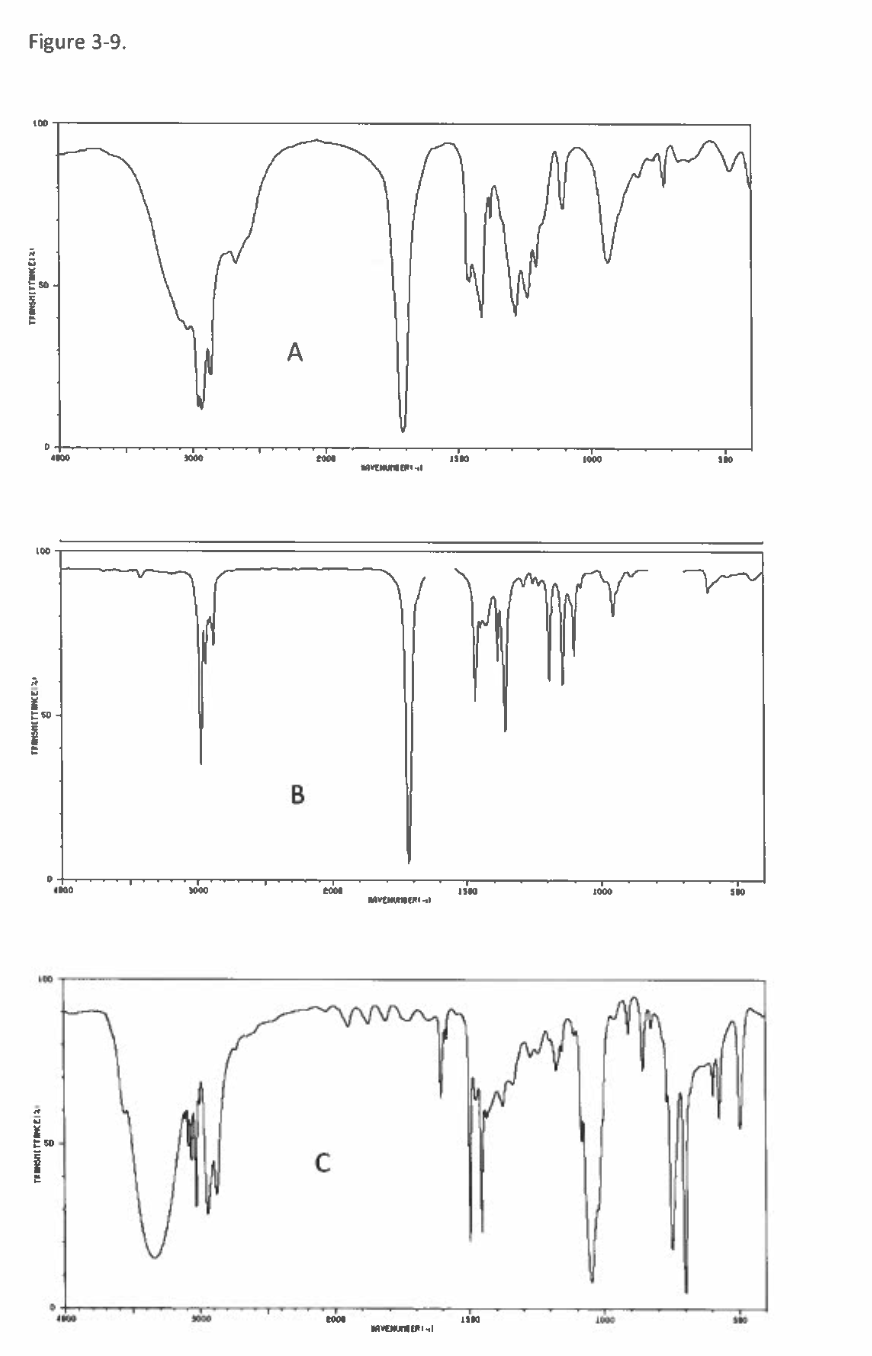
![In Figure 3-9. Match the spectrum to it's functional group.
A
B
✓ [Choose ]
ketone or ester
aldehyde
carboxylic acid
alcohol
alkyne
[Choose ]
◄►](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F34a9c4cb-86d3-4919-ad5f-37407f4c9cd7%2Fed97c0e4-3b64-49e4-bac8-16c2c785db7f%2Fdm0j9uc_processed.png&w=3840&q=75)
Transcribed Image Text:In Figure 3-9. Match the spectrum to it's functional group.
A
B
✓ [Choose ]
ketone or ester
aldehyde
carboxylic acid
alcohol
alkyne
[Choose ]
◄►
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

