Fill in the blanks: An environmental chemist analyzed the effluent from an industrial process known to produce the compounds carbon tetrachloride (CCI4) and benzoic acid (HC7H502), a weak acid that has one acidic hydrogen atom per molecule. A sample of this effluent weighing 0.3518 g was shaken with water and the resulting aqueous solution required 10.59 mL of 0.1546 M NaOH for neutralization. Calculate the mass percent of HC7H5O2 in the sample.
Fill in the blanks: An environmental chemist analyzed the effluent from an industrial process known to produce the compounds carbon tetrachloride (CCI4) and benzoic acid (HC7H502), a weak acid that has one acidic hydrogen atom per molecule. A sample of this effluent weighing 0.3518 g was shaken with water and the resulting aqueous solution required 10.59 mL of 0.1546 M NaOH for neutralization. Calculate the mass percent of HC7H5O2 in the sample.
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter3: Chemical Reactions
Section: Chapter Questions
Problem 150QRT
Related questions
Question
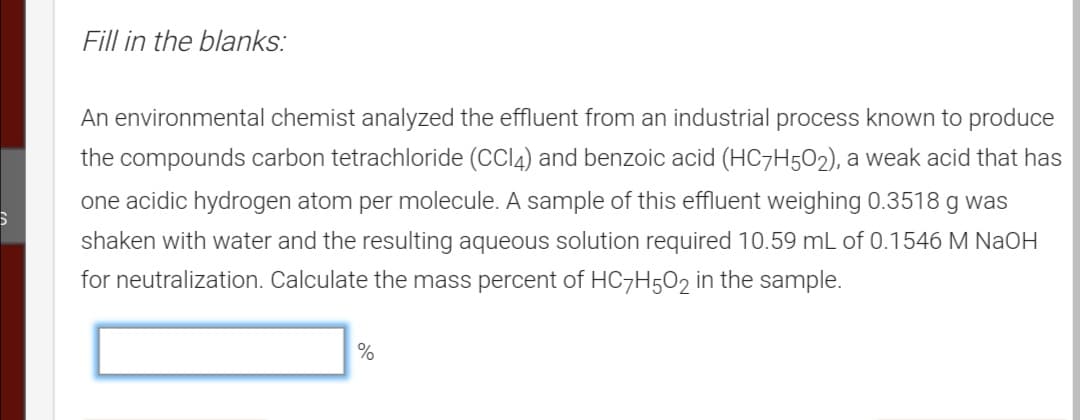
Transcribed Image Text:Fill in the blanks:
An environmental chemist analyzed the effluent from an industrial process known to produce
the compounds carbon tetrachloride (CCI4) and benzoic acid (HC7H502), a weak acid that has
one acidic hydrogen atom per molecule. A sample of this effluent weighing 0.3518 g was
shaken with water and the resulting aqueous solution required 10.59 mL of 0.1546 M NaOH
for neutralization. Calculate the mass percent of HC7H5O2 in the sample.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning