Five carbohydrate samples were analyzed for their qualitative reactions and the results are summarized in Table 1 below. Match the reaction profile of each sample to their possible identity from the choices.
Five carbohydrate samples were analyzed for their qualitative reactions and the results are summarized in Table 1 below. Match the reaction profile of each sample to their possible identity from the choices.
Chapter16: Adult And Pediatric Dosages Based On Body Surface Area
Section: Chapter Questions
Problem 7.1P
Related questions
Question

Transcribed Image Text:CARBOHYDRATE REACTIONS
Five carbohydrate samples were analyzed for their qualitative reactions and the results are
summarized in Table 1 below. Match the reaction profile of each sample to their possible identity from
the choices.
Table 1. Qualitative Tests of Carbohydrate Samples
Barfoed's Test
Rusty precipitates
Aqua blue color
Rusty precipitates
Aqua blue color
Aqua blue color
Seliwanoff's Test
Pale yellow solution
Pale yellow solution
Cherry red solution
Pale yellow solution
Pale yellow solution
Benedict's Test
Rusty precipitates
Aqua blue color
Rusty precipitates
Aqua blue color
Rusty precipitates
Lugol's Test
Brown color
Brown color
Brown color
Blue-brown color
Brown color
Sample No.
1
4
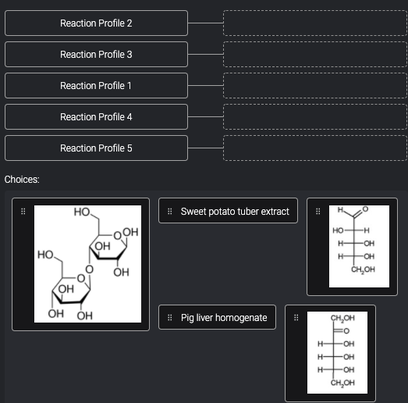
Transcribed Image Text:Reaction Profile 2
Reaction Profile 3
Reaction Profile 1
Reaction Profile 4
Reaction Profile 5
Choices:
HO
! Sweet potato tuber extract
H.
OOH
он
HO
H-
H-
-OH
но.
H-
OH
OH
CH,OH
OH
ÓH ÓH
H
# Pig liver homogenate
CH,OH
H-
-O-
H-
-OH
H-
OH
CH,OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you


Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage



Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage

