Q: Consider the E2 elimination of 3-bromopentane with hydroxide. :Br CH3 product + Br+ H2O H CH3CH2…
A: E2 mechanism is a bimolecular elimination method. It is also known as concerted mechanism because…
Q: Complete the reaction scheme below with the missing structure and any other necessary reagents. You…
A: The answer is as follows:
Q: Hg2+ Но c=CH2 H3C H3C-C=C-H H30
A:
Q: Draw the structures for the intermediates and product (left to right) as represented by A to C in…
A: In this question we have to tell the product of the reaction sequence.
Q: HC=CH The synthesis above can be performed with some combination of the reagents listed below. Give…
A: For deprotonation of acetylene we need strong base such as NaNH2.
Q: Q17. Compound 1 can undergo an intramolecular reaction to give cyclic product 2. Using curly arrows,…
A: Cyclic product is formed , which is cyclic amide , ester functional group act as electrophile and…
Q: 1. Orbitals and stereochemistry in the E2 reaction a. Add the orbitals and missing substituent…
A: E2 : it is a bimolecular elimination reaction . it happens in a concerted path where a strong base…
Q: 4. For the following reaction scheme, please convert the starting material to the corresponding…
A: The reactions taking place are given as,
Q: 1. Provide the missing starting material, reactant or product. Show appropriate stereochemistry. он…
A:
Q: Draw the major organic products of the reactions, showing stereochemistry as needed. If no…
A: The details solution for this reaction is provided below in attach image.
Q: CH3H ff. H ÓH + CEN with pyridine in DMSO Reaction Type(s) + Mechanism(s) Product(s).…
A: The answer is given as follows
Q: 4. The following is an intramolecular Sn2 reaction. (Although not technically Sn2 because it is…
A: See answer below.
Q: Draw the product of the reaction shown below. Use wedge and dash bonds to indicate relative…
A: The product is given below:
Q: CH3 H3C- CH- CH -CH, OH HSO, Reaction Type(s) a. Mechanism(s) Product(s). Stereochemistry (if…
A: ***Since you have posted multiple independent questions in the same request, we will solve the first…
Q: te a full mechanism for each reaction shown below. (I) Clearl (iii) show stereochemistry where…
A: According to our guideline, I can answer only the first three sub-parts.
Q: n each case below select the synthetic procedure that could be used to carry out the transformation…
A: The product of the following reaction method are shown as follow.
Q: Give a clear handwritten mechanism and product for given below Sn2 reaction..a reaction shows all…
A: SN2 is reaction mechanisms:
Q: Draw the product of the reaction shown below. Use wedge and dash bonds to indicate relative…
A:
Q: 6. Determine whether each reaction occurs by an Sn2, Sn1, E1, and/or E2 reaction. Provide a detailed…
A: Hello. There are multiple subparts to this question. The first three subparts have been solved. To…
Q: CI КОН ELOH H Нeat
A:
Q: H;C CH3 KOC(CH 33 .CI Solvent: Toluene -CH3 CH3
A: Reaction of alkyl halide produce substitution product with strong nucleophilic
Q: En ontain viruses, Unless you need to edit, it's safer to stay in Protected View. 1. Write the…
A: The solutions are given below -
Q: Draw the complete mechanism of the ring opening of the epoxide below with methanol under both acidic…
A:
Q: Consider the E1 reaction of 3-bromo-2-cyclohexyl-butane. How many elimination products are…
A:
Q: Consider the elimination reaction shown below: Me- H2SO4 Ме— он Me alkene-containing product(s)…
A: When alcohols are treated with Conc. sulphuric acid, they undergo dehydration i.e. removal of water…
Q: Provide a curved-arrow mechanism for the following reaction. Be sure to use your mechanism and a few…
A: So, in this reaction, the alkene acts as a nucleophile, attacking the electrophilic bromine, giving…
Q: Use curved arrows to show how E is converted to F by a two-step reaction sequence consisting of a…
A: In sigmatropic rearrangement, the rearrangement of pi bond and the breaking of sigma bond take…
Q: 6. Determine whether each reaction occurs by an SN2, SN1, E1, and/or E2 reaction. Provide a detailed…
A: → In above base is weak so it show SN2 reaction.
Q: please answer in detail
A: Secondary alcohols when treated with potassium dichromate and sulfuric acid, they gets oxidized to…
Q: Chemistry 3. Complete the following reaction scheme. Give all product(s) and indicate major or minor…
A:
Q: Complete the reaction scheme below with the missing structure and any other necessary reagents. You…
A: The answer is as follows:
Q: CHa Br, FeBr, NHCOCH, heat H3C CH, SO,, H-SO, Br CF3 heat
A:
Q: 3. Explain, by means of a detail mechanism for each, why the two reactions give the same product.…
A: ->In presence of base elimination reaction occur and alkene formed.it removed antiperiplanar…
Q: Use curved arrows to show how E is converted to F by a two-stepreaction sequence consisting of a…
A: The reaction involves [1,5] sigmatropic rearrangement and [4+2] cycloaddition. A C-H bond is broken…
Q: Complete the reactions given below, write down the type of mechanism (SN1, SN2, E1, E2
A: SN1 and SN2 reaction require weak nucleophile and strong nucleophile.
Q: #4 - 1-Methylcyclohexene, shown in the box below, is a versatile starting material for a variety of…
A: The reactions taken are addition reaction and hydroboration oxidation.
Q: For the dehydrohalogenation (E2) reaction shown, draw the major organic product, including…
A: E2 reaction is a bimolecular elimination reaction to give an alkene. In the transition state, the…
Q: Draw mechanism arrows and then predict the major products and stereochemistry. Also include the…
A: The given compound, 3,4-dimethylhex-3-ene is a symmetrical alkene. Thus, on hydroboration oxidation,…
Q: Draw the products of the two step reaction sequence shown below. Use dash and/or wedge bonds to…
A:
Q: Complete reaction with details and name the major and minor products. stereochemistry shoudl be…
A: We have to draw the product of bromination and chlorination
Q: In the formation of the following ether, which reaction is preferred and why? NOC NEOCH, CH,OH O…
A: The reactions given are,
Q: Each heterolysis step on the left does not readily occur, but the corresponding one on the right…
A: The given reactions undergoes : 1). SN1 (unimolecular nucleophilic substitution) reaction. And 2).…
Q: Could the reactions shown here take place by a concerted mechanism?
A:
Q: Show the El mechanism for the following reaction, which includes a rearrangement. Use your work to…
A:
Q: The first of the two heterolysis reactions below takes place readily, but the second one does not.…
A: Heterolysis reactions involves the breaking of bond in which two electrons in a bond distributed…
Q: If Zaitsev's rule does not apply and the base is bulky then how will the product look?
A: Bulky base like potassium acetate will abstract beta proton from less hindered carbon to form less…
Q: Add curved arrows to draw the first part of the mechanism: tautomerization of a ketone to an enol.…
A: A mechanism of an organic reaction is shown by the movement of electrons. An arrow is used to show…
Q: Draw the major organic product generated in the reaction below. Pay particular attention to 1)…
A:
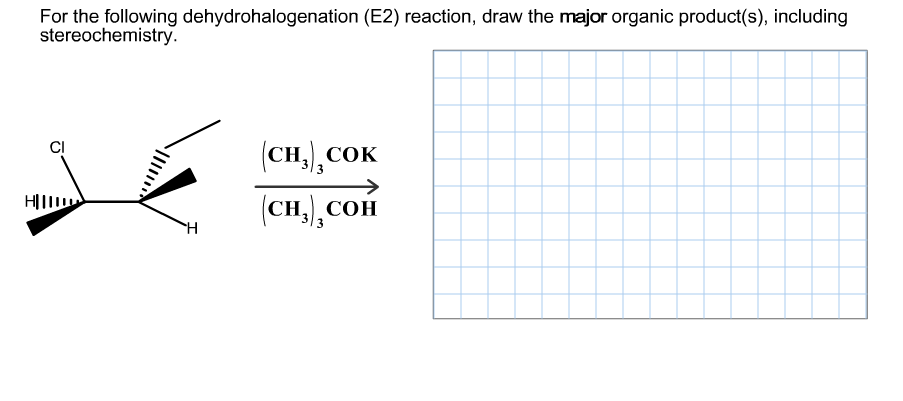
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 4 images

- In the box to the left of each reaction below, write the mechanism by which it occurs (could be SN1, SN2, or E1, or even 2 of them). Then draw the product(s).3) Explain how and why the reaction below results in the observed regiochemistryand/or stereochemistry.Can you do 3 and 4 and explain it? And how yo identify it. State the mechanism in which through reaction proceeds sn2 or sn1
- Complete the following reactions with the majority products and the missing reaction conditions. Indicate stereochemistry if applicable. Also indicate the type of reaction that took place (SN1, SN2, E1, E2, E1bc)Draw all possible alkenes that could be generated from an E2 elimination reaction of 3-bromo-3-methylhexane. Suggest conditions that can be used to make just one of the alkenes in a controlled manner (i.e., conditions that would lead to formation of just one alkene as the major product).Write whether it will be E1, E2, SN1, and/or SN2 Show the mechanism(s) Write the MAJOR product(s) State the stereochemistry of the product(s), when applicable Please do them how i set them up.. that the only way I understand.
- Predict the product, draw the mechanism, and plot the reaction coordinate diagram for theE1 reaction.Provide the mechanism for the conversion shown below. CH3Li is effectively H3C-Li+. What type of mechanism is occuring?Explainwhythefollowingdeuterated1-bromo-2-methylcyclohexaneundergoes dehydrohalogenation by the E2 mechanism, to give only the indicated product. Two other alkenes are not observed.
- Between E1 and E2, which reaction mechanism is most efficient to synthesize the (E) stereoisomer of this product on an industrial scale?Draw the arrow-pushing mechanism and propose the products. When appropriate, make sure to indicate the appropriate stereo-/regio-chemistry of the product(s). Note that the mechanism(s) by which the product(s) form in reactions A and B are shown in pink. For Reactions C, D, and E you need to deduce the mechanism by carefully analyzing yout reagents and conditions.Please help with the following ochem mechanisms.... 1. Provide the stepwise mechanisms for the following reactions (see attached picture)

