Force Completion Once started, this test must be completed in one sitting. Do not leave the test before clicking Save and Submit Remaining Time: 36 minutes, 58 seconds. Question Completion Status: A Moving to another question will save this response. Question 9 Which of the following compounds gives an infrared (IR) spectrum with a peak at 1730 cm (strong peak)? ОН ОН 1 2 3 4 2 3 4 A Moving to another question will save this response. MacBook Air OO0 D00 F4 F2 II F8 F3 F5 F6 F7 F9 $ & 2 5 7 8 tA4 #3
Force Completion Once started, this test must be completed in one sitting. Do not leave the test before clicking Save and Submit Remaining Time: 36 minutes, 58 seconds. Question Completion Status: A Moving to another question will save this response. Question 9 Which of the following compounds gives an infrared (IR) spectrum with a peak at 1730 cm (strong peak)? ОН ОН 1 2 3 4 2 3 4 A Moving to another question will save this response. MacBook Air OO0 D00 F4 F2 II F8 F3 F5 F6 F7 F9 $ & 2 5 7 8 tA4 #3
ChapterU5: Fire: Energy , Thermodynamics, And Oxidation-reduction
SectionU5.116: How Absorbing: Spectroscopy
Problem 7E
Related questions
Question
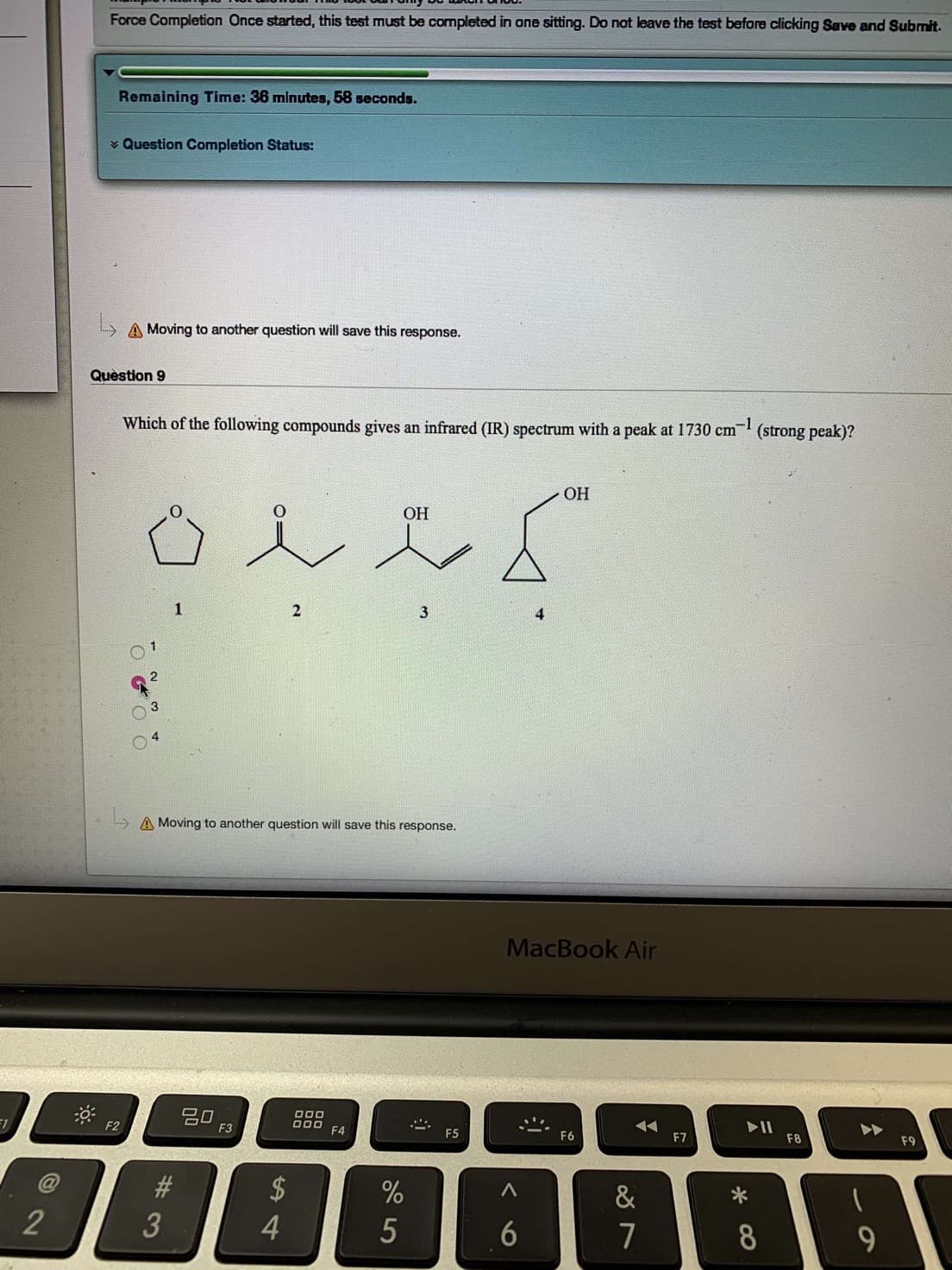
Transcribed Image Text:Force Completion Once started, this test must be completed in one sitting. Do not leave the test before clicking Save and Submit
Remaining Time: 36 minutes, 58 seconds.
Question Completion Status:
A Moving to another question will save this response.
Question 9
Which of the following compounds gives an infrared (IR) spectrum with a peak at 1730 cm
(strong peak)?
ОН
ОН
1
2
3
4
2
3
4
A Moving to another question will save this response.
MacBook Air
OO0
D00 F4
F2
II
F8
F3
F5
F6
F7
F9
$
&
2
5
7
8
tA4
#3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning