Q: Consider the decomposition reaction: XY2(s) X(g) + 2 Y(g) where X and Y are some chemical groups. At…
A: In the equilibrium constant expression, the concentration of pure solids and liquids are considered…
Q: Please do not round off intermediate calculations. Thank you.
A:
Q: Consider data from the table of standard reduction potentials for common half-reactions, in your…
A: The anodic half -reaction that would produce a spontaneous reaction of Sn4+ to Sn2+ but not Sn2+ to…
Q: What is the solubility of arsine (in units of grams per liter) in water at 25 °C, when the AsH3 gas…
A:
Q: Which of the following would successfully perform the shown reaction? (A) (B) (C) OH 1) NaH 2) EtBr…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: estion 19 of 20 The pOH of a basic solution is 2.65. What is pH?
A: The question is based on the concept of pH of the solution. it is defined as a negative…
Q: For the chemical in the reaction below, ΔHf° is zero for __________. 2Co (s) + H2 (g) + 8PF3 (g) →…
A:
Q: b How many kinds of chemically non-equivalent carbons are there in each of the following compounds?…
A: Atoms reflected by mirror plane are called equivalent. Atom bisected by mirror plane are called…
Q: - N₂(9)+ 3H₂(g) → 2NH₂(9) At the temperature the engineer picks, the equilibrium constant K, for…
A: Answer: Relation between reaction quotient and equilibrium constant is shown below: 1. When QP=KP…
Q: 10. What is the major product in the following reaction? of A. I 1. NaOEt, EtOH, 25 °C do ď B. II 2.…
A:
Q: tarting with any alcohols (SIC) and any inorganic reagents required make the target molecule. Show…
A: Concept introduction: Retrosynthesis analysis is an important tool for Designing any organic…
Q: Determine the value of Ksp for Cu(103)2 by constructing an ICE table, writing the solubility…
A:
Q: Calculate the change in pH when 7.00 mL of 0.100 M HCl(aq) is added to 100.0 mL of a buffer solution…
A: To solve this problem, we need to first determine the initial pH of the buffer solution. The buffer…
Q: What happens when HCI is added to a buffer composed of CH3COOH and CH3COO™? (A) There is no reaction…
A: Buffer is the combination of two component. 1- a weak acid and its salt from a strong base - acidic…
Q: Choose the right completed variant of the following acid-base reactions. → Na2SO4 (aq) + 2H₂O(1) In…
A: Balanced chemical equation: Balanced chemical equation can be define as the reaction in which number…
Q: Gaseous hydrogen iodide is placed in a closed container at 425 ∘C ∘ C , where it partially…
A: The equilibrium constant (K) is given by the ratio of the products of the molar concentration of…
Q: Carbon monoxide and water vapor are formed when carbon dioxide and hydrogen react. CO2(g) + H2(g) ⇌…
A:
Q: In the reaction NH3(aq) + H₂O(1)⇒ NH4+ (aq)+ OH(aq) does water act as a Brønsted-Lowry acid, a…
A: A substance which donates a proton or H+ ion to the other compound and forms a conjugated base is…
Q: Be sure to specify states such as (aq) or (s). If a box is not needed leave it blank. Write the net…
A:
Q: After spilling battery acid in your garage, you need to neutralize it with a strong base. Which of…
A: Please find your solution below : pH of a solution is the measure of how acidic or basic a substance…
Q: Why is B and D not a meso compound like A and C
A: The compound in which more than one chiral center in present and there should be one plane of…
Q: I II III IV V
A: Free radicals are electron deficient species and hence any factor which increase electron density…
Q: An alien cell membrane at 41.000°C is found to be permeable to Al³+ but not to anions, and analysis…
A: Given: Temperature = 41.000oC Cell membrane is permeable to[Al3+] but not to anions Inside…
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: The unbalanced redox reaction given is ClO2 + Cr3+ → ClO3− + Cr2+
Q: Complete the equation for the following acid-base reaction by drawing a structural formula for the…
A: We have been given two incomplete reactions.We have been asked the missing reactant in one organic…
Q: Calculate the standard reaction entropy for N2(g) + 3 H2(g) → 2 NH3(g) at 25 °C. (b) What is the…
A:
Q: ОН CO H Ac2O H₂SO4 (cat) ?
A:
Q: b. Ss (s) + F2 (g) + SF6 (g) (be careful about the phases) S8 +24F₂,85 Fo [SF] [S₂] [F₂ ] ²4 Qc =
A: Given Equilibrium Equation, S8(s) + 24 F2 (g) <-------> 8 SF6(g) We need to write the value…
Q: Please do not round off intermediate calculations. Thank you.
A: Given that : The mass of lead nitrate is 26.4g. The volume of ammonium sulfate solution is 150 mL.…
Q: What is the major product of the following sequence of reactions? 1. NaOEt, EtOH 2. H₂O+ 3. 0= Br 4.…
A:
Q: Determine the molar solubility with the generic formula AB2 if ksp=2.56*10^-2
A:
Q: Suppose 3.32 g of nickel(II) iodide is dissolved in 200. mL of a 0.30 M aqueous solution of…
A: The equation is,
Q: The H-NMR spectrum for the product shows two doublets at 7.09 ppm and 7.75 ppm, with equal…
A: We need to give reason 'why doublet due to Hd is at larger chemical shift value than doublet due to…
Q: draw the structures of the geometric and optical isomers of the compound [Co(gly)3
A: We have been given an inorganic compound [Co(gly)3].We have been asked to draw the structures of…
Q: Consider the decomposition reaction: X(g) + 2 Y(g) XY2(s) where X and Y are some chemical groups. At…
A: Answer: Value of equilibrium constant is equal to the ratio of molar concentration of products and…
Q: Light fuel oil with an average chemical composition of C10H18 is burned with oxygen in a bomb…
A: This problem is based on Thermodynamics. The solution is given below.
Q: Open bodies of water can act as carbon sinks due to the ability of carbon dioxide to dissolve in…
A: Please find your solution below :
Q: Suggest an efficient synthesis for the following transformation: A-4x The synthesis above can be…
A: Ozonolysis of the alkene produces carbonyl compounds. It has two step, in step one ozonide…
Q: In classroom demonstration, a teacher pours a supersaturated solution of sodium acetate and water…
A: Given In classroom demonstration, a teacher pours a supersaturated solution of sodium acetate and…
Q: alculate ASn for the balanced chemical equation: ASixn ΑΣΦ -6.56-104 2H₂S(g) + 302(g) → 2H₂O(g)…
A:
Q: Draw the products of each acid-base reaction. EXAMPLE: a. b. CH3CH₂ propanoic acid OH + CH3CH₂CH₂ OH…
A:
Q: Problem 18.18 What starting materials are needed to prepare each alkene by a Wittig reaction? When…
A:
Q: The molar concentration of H3O+ in pure water at 25 °C is: (A) 0.0 M (B) 10-¹4M (C) 10-7M (D) 10 M…
A: We know that the pure water at 25 °C is neutral and it's pH and pOH value is equal to 7 => pH =…
Q: 2. When the following pair of reactants are combined in the presence of a basic catalyst, a number…
A: in presence of base, carbonyl compound having enolisable alpha hydrogens, undergo aldol condensation…
Q: Medical grade Heliox, used in cases of respiratory distress, may contain 20.0 mole % oxygen (0₂) and…
A:
Q: Use the References to access important values if needed for this question. Does a reaction occur…
A: Ionic equation is the chemical equation in which the dissolved ionic compounds are written as…
Q: CH3 ∞ ENCH₂CH3
A: For retro-synthetic analysis, we have to select a particular bond , so that breaking of that bond we…
Q: a. On the basis of the general solubility rules, choose the substance that is not likely to be…
A: The solubility rules are given below. 1. Most nitrate salts are soluble 2. Most salts of Na+, K+,…
Q: At what ph will Al(OH)3 precipitate if Al 3+ =3.10x10^-4 M ksp = 1.9x10^-33
A: The precipitation of Al(OH)3 occurs when the product of the ion concentrations of Al3+ and OH-…
Q: A radioactive isotope has a half life of 16.9 minutes. How long will it take for the radiation from…
A:
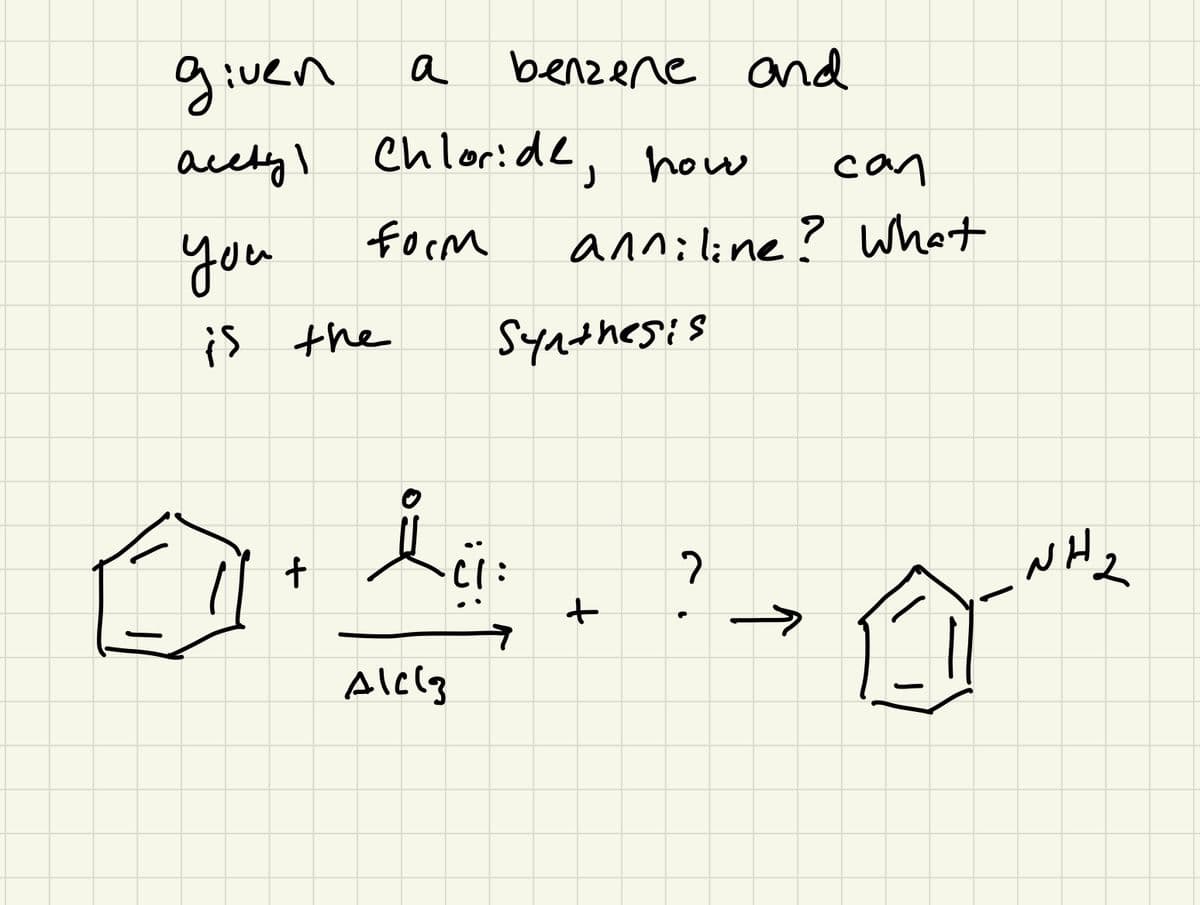
Please see photo
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Glucosamine, one of the eight essential monosaccharides (Section 25-7), is biosynthesized as its 6-phosphate derivative from fructose 6-phosphate by reaction with ammonia. Propose a mechanism.Identify what compound/s are non-reducing sugarDraw the organic product(s) formed when CH3CH2CH2OH is treated withfollowing reagent : POCl3, pyridine

