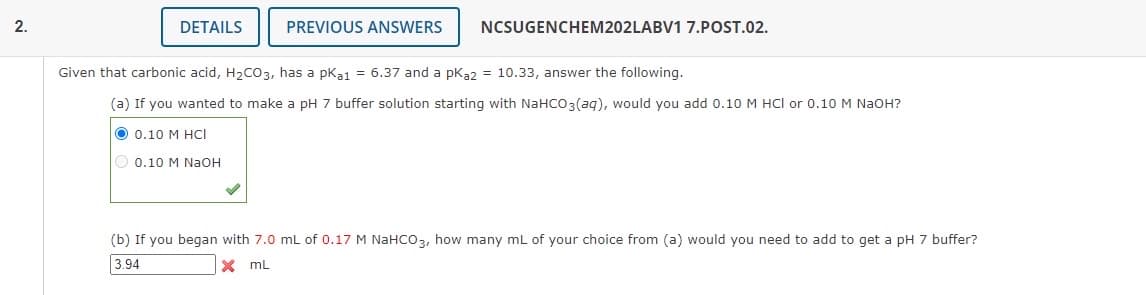
Given that carbonic acid, H₂CO3, has a pka1 = 6.37 and a pka2 = 10.33, answer the following. (a) If you wanted to make a pH 7 buffer solution starting with NaHCO3(aq), would you add 0.10 M HCl or 0.10 M NaOH? 0.10 M HCI 0.10 M NaOH (b) If you began with 7.0 mL of 0.17 M NaHCO3, how many mL of your choice from (a) would you need to add to get a pH 7 buffer? 3.94 X mL
Given that carbonic acid, H₂CO3, has a pka1 = 6.37 and a pka2 = 10.33, answer the following. (a) If you wanted to make a pH 7 buffer solution starting with NaHCO3(aq), would you add 0.10 M HCl or 0.10 M NaOH? 0.10 M HCI 0.10 M NaOH (b) If you began with 7.0 mL of 0.17 M NaHCO3, how many mL of your choice from (a) would you need to add to get a pH 7 buffer? 3.94 X mL
Chapter14: Principles Of Neutralization Titrations
Section: Chapter Questions
Problem 14.38QAP
Related questions
Question
Transcribed Image Text:2.
DETAILS
PREVIOUS ANSWERS NCSUGENCHEM202LABV1 7.POST.02.
Given that carbonic acid, H₂CO3, has a pka1 = 6.37 and a pka2 = 10.33, answer the following.
(a) If you wanted to make a pH 7 buffer solution starting with NaHCO3(aq), would you add 0.10 M HCl or 0.10 M NaOH?
O 0.10 M HCI
0.10 M NaOH
(b) If you began with 7.0 mL of 0.17 M NaHCO3, how many mL of your choice from (a) would you need to add to get a pH 7 buffer?
3.94
XML
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

