How many grams of AgCI will be formed when 60 mL of 0.500M AGNO, is completely reacted according to the balanced chemical reaction: FeCl,(aq) + 3 AGNO,(aq) - 3 AGCI(s) + Fe(NO,)2(aq) STARTING AMOUNT
How many grams of AgCI will be formed when 60 mL of 0.500M AGNO, is completely reacted according to the balanced chemical reaction: FeCl,(aq) + 3 AGNO,(aq) - 3 AGCI(s) + Fe(NO,)2(aq) STARTING AMOUNT
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter15: Solutions
Section: Chapter Questions
Problem 39QAP: 39. Standard solutions of calcium ion used to test for water hardness are prepared by dissolving...
Related questions
Question
Please use dimensional analysis and only use the numbers that are available in this picture. note that 0.03 is not an option.
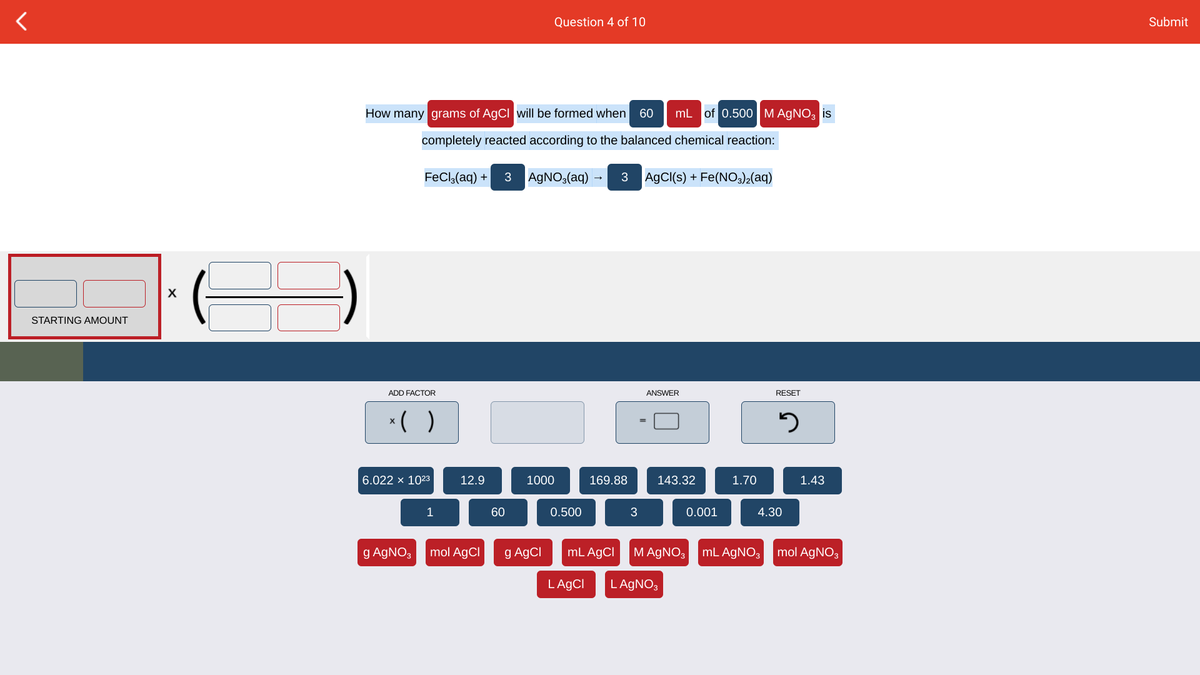
Transcribed Image Text:Question 4 of 10
Submit
How many grams of AgCI will be formed when 60| mL of 0.500 M AGNO, is
completely reacted according to the balanced chemical reaction:
FeCla(aq) +
3
AGNO3(aq)
3 AgCI(s) + Fe(NO3)2(aq)
X
STARTING AMOUNT
ADD FACTOR
ANSWER
RESET
*( )
6.022 x 1023
12.9
1000
169.88
143.32
1.70
1.43
1
60
0.500
0.001
4.30
g AGNO3
mol AgCl
g AgCI
mL AgCI
M AGNO3 mL AGNO3
mol AGNO3
LAGCI
L AGNO3
Expert Solution
Step 1
The balanced reaction taking place is given as,
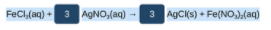
Given : Concentration of AgNO3 = 0.500 M
And volume of AgNO3 solution = 60 mL = 0.060 L (since 1 L = 1000 mL)
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning