Q: How many atoms of Al are needed to react with 34.5 grams of O2 gas?
A: Introduction: Stoichiometry is a process that used to calculate amounts in reactions by using the…
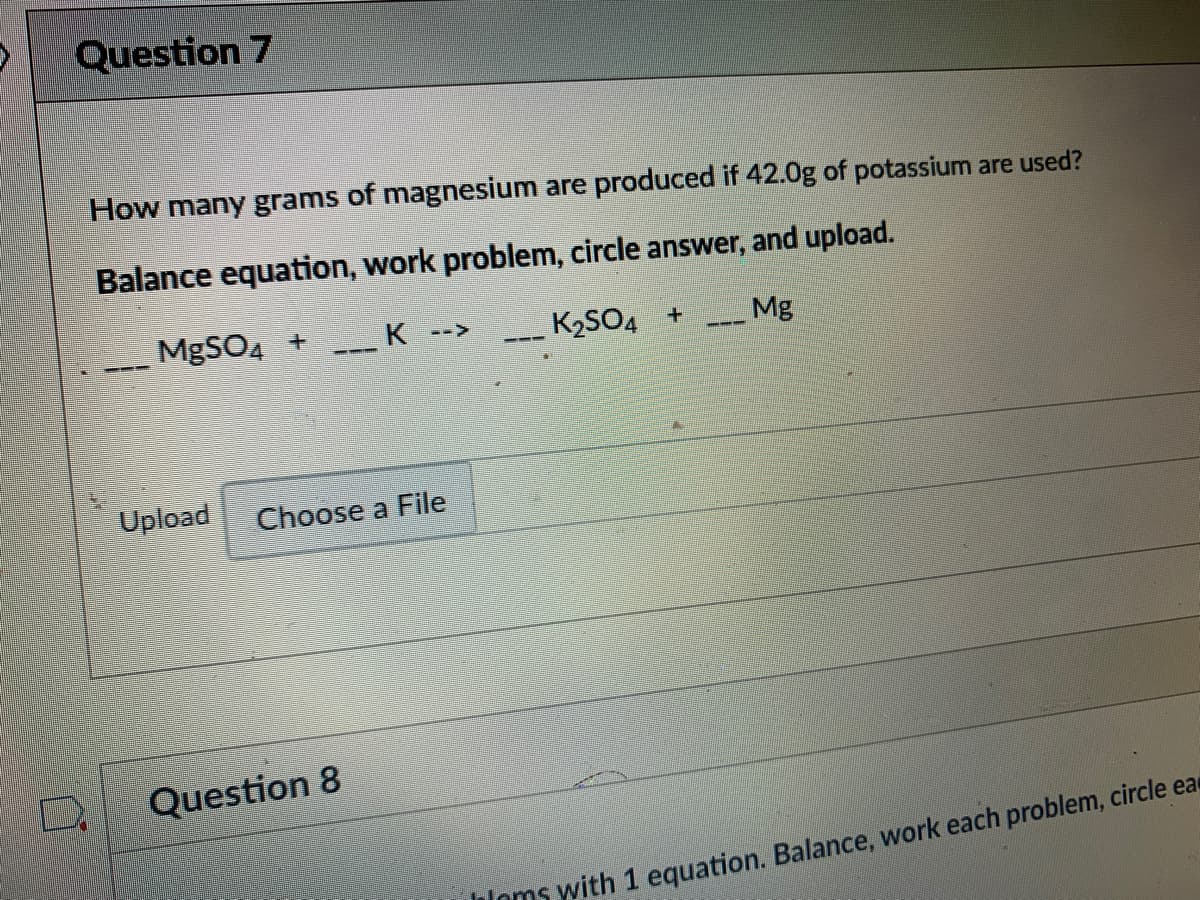
Q: how many moles of ammonia can be oxidized by 303 grams of oxygen?
A:
Q: how many grams of potassium nitrate(101.1g/mol) must be heated for decomposition to produce 88.6kg…
A: Molar mass of potassium nitrate = 101.1 g/mol Mass of produced oxygen = 88.6 kg Mass of potassium…
Q: A sulfur containing compound is treated chemically to convert all its sulfur into Barium sulfate…
A:
Q: 5.0g of solid zinc carbonate are heated to produce oxide and carbon dioxide. (a) Write a balanced…
A: Number of moles of a chemical compound is used to calculate the amount of the compound in the given…
Q: How many pounds of calcium fluoride rock is needed to produce 3000 Ibs of a 60% HF acid per day if…
A: A numerical problem based on quantitative analysis, which is to be accomplished.
Q: How many moles of carbon dioxide are needed to consume 5.2 moles of oxygen?
A: The answer to the following question is given as:
Q: How many moles of aluminum oxide can form if 26.2 moles of Al metal react with an unlimited amount…
A: The reaction taking place is Al (s) + O2 (g) -----> Al2O3 (s) Balancing : Since we have 3 O in…
Q: How many grams of sodium are required to react with 0.800mol of oxygen?
A:
Q: C 4H 8 +6O2 4CO2 + 4H 2OWhat is the stoichiometric ratio of H 2O to O 2? (Include units.)
A: Combustion reaction is the type of reaction of a hydrocarbon with oxygen gas. For hydrocarbons,…
Q: Calculate the theoretical yield of lead fro
A:
Q: Suppose an aqueous solution containing 1.00 g of lead(II) acetate is treated with 5.90 g of carbon…
A: The balanced reaction between lead acetate and carbon dioxide is as shown below. Pb(CH3COO)2 + CO2…
Q: How many moles of oxygen would be produced by the decomposition of 75.0g of copper (II) nitrate?
A: Two moles of copper (II) nitrate undergoes decomposition to form one mole of oxygen gas according to…
Q: 6. How many moles of calcium will react with 2.00 grams of:
A: the balanced equation of Ca with each acid is given below a. Ca + 2 HCl -------- > CaCl2 +…
Q: When the equation ? PbS+? O2 →? PbO+? SO2 is balanced, the coefficients are
A: A chemical equation is the representation of a chemical reaction. In chemical equations, the…
Q: When the reaction between sodium hydroxide and sulfuric acid is balanced, the stoichiometric…
A: The chemical equation for the reaction of sodium hydroxide and sulfuric acid sis represented as…
Q: Calculate the theoretical yield(Twice the moles of Cu Foil), Calculate the moles of CuCl Prepared…
A: Mass of copper foil = 1.0762 g Moles of copper foil = 0.016935 moles
Q: How much zinc chloride could be made from 1 mole of Zn and 1 mole of HCl if the reaction is?
A: The given stoichiometric equation is Zn(s) + 2 HCl (aq) → ZnCl2(aq) + 2 H2(g) 1mol…
Q: How many molecules of magnesium hydroxide are produced from 0.960 mole of magnesium oxide?
A: One mole of compound contains Avogadro number of its molecules. Avogadro Number = 6.022 × 1023
Q: How many moles of silver chromate will be produced from 143 g of silver nitrate?
A: Number of moles of a substance is given by the ratio of mass of the substance to that of the molar…
Q: 5. How many grams of oxygen can be produced from the decomposition of 100.0 g of KCIO,?
A: Question 5) Given, Mass of KClO3 decomposes = 100.0 g Mass (in grams) of Oxygen gas produced = ?…
Q: How many grams of chlorine gas can be produced from theelectrolytic decomposition of 874 g of…
A: Given data contains, Mass of calcium chloride is 874g. Molar mass of calcium chloride is…
Q: How many grams of phosphorus is need if 1.06 kg of oxygen was converted toP4O10
A: Chemical reaction : 4 P + 5 O2 ---> P4O10 Mass of oxygen : 1.06 Kg 1 kg = 1000 g 1.06 Kg =…
Q: How many grams of O₂ can be prepared from the thermal de-composition of 4.27 kg of HgO? Name and…
A: Given: Mass of HgO decomposed = 4.27 kg
Q: What mass of ammonia is consumed by the reaction of 4.8 g of oxygen gas?
A: number of moles = mass/molar mass molar mass of ammonia is 17.0305 g/mol molar mass of O2 is 31.999…
Q: how many grams of limestone must be reacted to produce 500 grama of lime
A: The Question is based on the concept of Reaction Stoichiometry. We have to determine how much…
Q: Given 92.45 g of CO2, what is the mass of the carbon?
A: Given 92.45 g of CO2, what is the mass of the carbon ?
Q: How many grams of product will be formed by completely burning 7.80 g of aluminum on excess oxygen?
A:
Q: How many O2 molecules are needed to react with 4.58g of S?
A: Number of molecules can be calculated by using following formula Number of molecules = moles ×…
Q: How many moles of potassium chlorate must react to produce 0.73 moles of oxygen?
A: The decomposition reaction of potassium chlorate is as follows: 2KClO3 → 2KCl + 3O2
Q: How many grams of nickel (III) chloride can be formed from 25.85 grams of solid nickel?
A: Recall the reaction to obtain nickel(III) chloride from solid nickel…
Q: How many grams of O2 will react completely with 35 grams of PH3?
A: The number of moles of a substance can be obtained by dividing its mass by molar mass. PH3 reacts…
Q: 0.0984g of an unknown chloride is dissolved in water a solution of silver nitrate is added an…
A: Weight of unknown chloride=0.0984g Weight of silver chloride=0.2290g
Q: If you burn 29.2 g of hydrogen and produce 261 g of water how much oxygen reacted
A:
Q: Consider the following reaction. How many moles of oxygen are required to produce 10.0 moles of…
A:
Q: Using the balanced chemical equation below, what mass of H3PO4 is formed given 41.36 grams of water…
A: Given that: Mass of water = 41.36 g To find: Mass of phosphoric acid formed =?
Q: How many grams of iron(II) nitrate will react with 0.24 grams of sodium phosphate?
A: Given data contains, Mass of sodium phosphate=0.24 grams Sodium phosphate reacts with iron (ii)…
Q: Calculate the theoretical yield given the balanced equation below and 50.0 g calcium nitrate…
A: According to the question, calcium nitrate reacts with potassium phosphate to produce calcium…
Q: How mant grams of Aluminum are required to produce 17.5 g of hydrogen
A: The reaction of Aluminum to produce Hydrogen is as follows: 2Al(s) +…
Q: How many moles of electrons are required to produce 18.1 g of potassium metal from a sample of…
A: We are hereby asked that how many moles of electrons are required to produce 18.1 g of potassium…
Q: How many grams of silver chloride can be made by reaction of 5.00 g of silver nitrate with 5.00 g of…
A: GivenMass of silver nitrate = 5gMass of aluminum chloride= 5g The equation for the given reaction is
Q: How many grams of water will be produced when 26.00 g of glucose reacts with 26.00 g of oxygen?
A:
Q: How many grams of nitrogen dioxide is produced if 14g of nitrogen and 25 grams of oxygen mix
A: To find the mass of nitrogen dioxide gas.
Q: How many moles of potassium would be necessary to produce 23.5g of potassium oxide?
A: Mass of potassium oxide produced = 23.5 g Here, we have to calculate the moles of potassium that are…
Q: Give the STOICHIOMETRIC TABLE for SO2+1/202_>SO3| need complete table clearly
A: With help of stoitiometric table we can set up equation for concentration initially and amiunt Left…
Q: if the reaction consumes 31.7 moles of ammonia how many grams of water would be produced
A: Note - Since you have not provided us with the reaction for the process, we will consider the…
Q: what is the percentage by mass of magnesium nitrate in
A: Given : Mass of solution =20.6 g Mass of Mg(NO3)2 = 2.91g To calculate = % by mass of…
Q: How many grams of H2 will be produced from 15.0 g of Al reacting with HCl? What mass of HCl is…
A: To solve this problem we will use the balanced chemical equation of the given reaction .
Q: If 250.grams of sodium azide undergoes decomposition, how many grams of sodium are produced?
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- GivenRaw Materials: 500 g calcium polysulphide and 1.5 kg hydrochloric acid Actual Yield: 343.4g precipitated sulfurFormulation: 250 mg per jarDosage form: Ointment Packaging: 100 jars per boxWhereas the balanced chemical reaction, as given in the problem is: CaS5+2HCl => 4S + CaCl2 + H2SMy problem: Find the number of dosage form and packaging that can be produced from the stoichiometric calculation based on the result upon finding the Mass to Mass Stoichiometry Calculation. I have based my answer to the mass stoichiometry from this link: https://www.bartleby.com/questions-and-answers/given-raw-materials-500-g-calcium-polysulphide-and-1.5-kg-hydrochloric-acid-actual-yield-343.4g-prec/b1de1102-f27f-4c8d-9b4e-c7fe8e6104b6Thanks, by the way. But can you still help me find the number of dosage from and packaging?Given: Active Ingredient: Potassium Nitrate Raw Materials: 3 kgs. acetic acid and 2.5 kgs. potassium bicarbonate Actual Yield: 1.66 kgs. potassium acetate Formulation: 1100 mg per 5 mL Dosage Form: Oral Solution Packaging 473 mL per bottle Synthesis and Packaging (Need answer)Balanced Chemical Equation: % composition by mass of each compound: Mass to Mass Stoichiometry Calculation: Limiting Reagent: Excess Reagent: Amount (g) in excess: % Yield: Number of dosage form and packaging that can be produced from stoichiometric solution:How many g of ZnS is needed to react completely with 37.72 gram O2 according to this equation?2ZnS + 3O2 --> 2ZnO + 2SO2 Put answer in box in regular , non-scientific, notation with 2 places past the decimal
- ll. Direction: Carefully read the following problemsDo the following :A. Balance the chemical equation B. Identify the limiting reactant and the excess reactant if applicable;C. Compute for the theoretical yield;D. Determine the percentage yield of the reaction;E. Calculate the percent error andF. Compute for the excess amount of the excess reactant if applicable;Given Active Ingredient: precipitated sulfur (ointment) Raw Materials: 500 g calcium polysulphide and 1.5 kg hydrochloric acid Actual Yield: 343.4g precipitated sulfur Formulation: 250 mg per jar Dosage form: Ointment packaging:100 jars per box Synthesis and Packaging (Need answer)- Balanced Chemical Equation:- % composition by mass of each compound:- Mass to Mass Stoichiometry Calculation:- Limiting Reagent:- Excess Reagent:- Amount (g) in excess: % Yield:- Number of dosage form and packaging that can be produced from stoichiometric solution:The following problem is patterned after Examples 6-7. The information is based on the spreadsheet shown below. Suppose Ingredient I is made up of 60% micoden and 40% water, Ingredient II is made up of 40% micoden, 40% bixon, and 20% water, and these ingredients are mixed together. What is the percentage of bixon in mixture a? Please type answer note write by hend
- Answer letter D, E and F only. ll. Direction: Carefully read the following problemsDo the following :A. Balance the chemical equation B. Identify the limiting reactant and the excess reactant if applicable;C. Compute for the theoretical yield;D. Determine the percentage yield of the reaction;E. Calculate the percent error andF. Compute for the excess amount of the excess reactant if applicable;4) NaCl (20.0 g) and AgNO3 (20.0 g) are dissolved in water and allowed to react (Reaction 2). I just need help with parts (d-f). PLease help me, question shown in photo Include phase labels where appropriate and balance all equations. I also included the work I've already done for context! (Molar masses (grams/mole): Na = 23.0; Cl = 35.5; Ag = 107.9; N = 14.0; O = 16.0) ????(??) + ????ଷ (??) → ? + ? RMass of weighing tray: 1.435 g Mass of weighing tray and salicylic acid: 3.469 g Mass of salicylic acid used: 2.027 Mass of filter paper: 0.862 g Volume of acetic acid anhydride used: 4.06 mL Drops of sulfuric acid used: 5 Data Table (Product Yield) Mass of watch glass: 16.342 g Mass of watch glass, acetylsalicylic acid, and filter paper: 19.609 g Actual yield: mass of acetylsalicylic acid: ? Calculate percent yield Show the calculation for the number of moles of salicylic acid present in the reaction mixture.
- I need help finding the therotical yield and the percent yield with the data provided: mass of filter paper crude= 0.184 g mass of filter paper+ crude product=1.211 g mass of filter paper pure=0.182 g mass of filter paper + pure product= 0.809 g melting point of pure product= 105-110Balance each equation by inserting coefficients as needed. P4O10+H2O-->H3PO4Show the balanced reaction and determine EM.

