Q: 35.6m to ft
A: formula for conversion of 1 meter to foot(ft)1 meter = 3.2808 foot
Q: Hello, thank you this was very helpful. However, i still dont completly understnad how to determin…
A: In the solution of formation of complex B and it's isomer, we can find the major product by Applying…
Q: подде НО N=C=N NH₂ Д.ОН
A: In the given reaction, the free carboxylic acid end of the peptide reacts with DCC and forms a good…
Q: Which of the number orders ranks the following molecules from strongest intermolecular forces to…
A: By considering given molecules and possible intermolecular forces of attraction we can arrange them…
Q: HCOOH (aq) + H2O (l) ⇋ H3O+ (aq) + HCOO- (aq) Suppose you wanted to increase the buffer capacity of…
A: Buffers are characterized by the pH range over which they can maintain more or less constant pH and…
Q: Draw the structural formulas of compounds A, C, D, E and F in the boxes provided above.
A: Structural formula definition.~ A structural formula display the atoms of the molecule in the order…
Q: An isotope of technetium (technetium-99m), has a half-life of 6 hours. A hospital has a sample which…
A: half-life(t1/2) of technetium-99m = 6 hours Initial amount [A]0 of technetium-99m = 36 mgWe need…
Q: 8. Cytosine can act as a base and accept a proton to form the charged species shown below. Label the…
A: the hydrogen bond donors(D) and acceptor (A) are label on the protonated cytosine. there are two…
Q: Syn. Rutile /UGS Chlorination Purification Electrolysis Chlorination: ____. + 2C+ Electrolysis:…
A: Answer :---- The complete reactions are :---- Chlorination :-- TiO2 + 2C + 2Cl2 -----> TiCl4 +…
Q: Aqueous sulfuric acid (H₂SO₂) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium…
A: Mass of H2SO4 = 7.8 gMass of NaOH = 4.7 gActual yield of Na2SO4 = 6.34 gPercentage yield of…
Q: The precursor of polyphenols is: Select one: НО НО OH "ОН
A: Here we have to find the correct one of the following which is the precursor of the polyphenols we…
Q: Select all the molecule pairs that are enatiomers to eachother A) O E) II... || *** G) None of the…
A: Enantiomers can be defined as one of two different isomers of a compound which is the mirror image…
Q: j) k) 1) Cl m) OCH3 مات OCH3 hv A hv
A: Pericyclic reaction proceeds based on the certain rules. These rules depends on the conditions like…
Q: If you carried out a one trial of an oxidation reaction in which citric acid was present and one…
A: Use of citric acid in foodCitric acid is often added to packaged food and drinks. It helps to keep…
Q: (short synthesis) bubong sill CI cheg of afwon siterünye singlesd (atalog 811 ale ste srl 26 gnol 26…
A:
Q: Br₂ Br 'Br
A: To draw the mechanism of the given organic reaction.
Q: Which of the following molecules is the major product of the reaction shown in the box? C
A: The reaction of benzene with acetyl chloride (CH3COCl), aluminum chloride (AlCl3), and water (H2O)…
Q: < Question 6 of 19 Solid sodium reacts with liquid water to form hydrogen gas according to the…
A:
Q: Circle and name at least three functional groups in the molecule below. Place an asterisk (*) beside…
A: In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the…
Q: Plutonium-238 is used in spacecraft to provide heat to power generators. One of these generators…
A: It is based on the concept of decay of the Radioactive substance .Here we are required find the mass…
Q: The synthesis of maleic acid anhydride (CH:O) can be accomplished by reacting benzene (C.H.) and…
A: 2 C₆H₆(l) + 9 O₂(g) → 2 C₄H₂O₃(s) + 4 CO₂(g) + 4 H₂O(g)mass of benzene (C₆H₆) reacts = 45.8 gmass of…
Q: Gaseous ethane (CH, CH₂) will react with gaseous oxygen (O₂) to produce gaseous carbon dioxide (CO₂)…
A:
Q: a. ? b. + HCI ?
A: In this question we have given two incomplete organic reactions and we have to complete those…
Q: In which of the following would silver chloride be most soluble? a 1M KNO3 b 1M HCI c 1M AgNO3 d 1M…
A:
Q: X Balance the chemical equation for each of the following: a. HCl(aq)+O₂(g) → H₂O(1)+ Cl₂(g) b.…
A: The balance chemical equation : has equal number of atoms on both sides of equation i.e. on the left…
Q: description that an atom subjected to a high voltage 1.1. suggests the becomes charged because it's…
A: The scientific method is defined as the process in which a scientist carries out a number of ways to…
Q: What mass of Fe(OH), would be produced by reacting 75.0 mL of a 0.0607 M Fe(NO3), solution with 125…
A:
Q: Is the following monosaccharide aldose or ketose? Using the language of the unit, justify your…
A: Monosaccharides are sugars i.e. polyhydroxy aldehyde or ketones . They cannot be hydrolysed into…
Q: What is the relationship between the electron configuration of an atom and its chemical reactivity,…
A: The electron configuration of an atom determines its chemical reactivity, which is closely tied to…
Q: H₂N Prostaglandin (PGE₂) -OCH3 OH
A: A functional group is a substituent or group of atoms in a molecule that causes the molecule's…
Q: What is the solubility of Mg(OH)2 at a pH of 10.90? (Ksp Mg(OH)2 is 1.6 × 10-1³)
A: The of pH of the solution = 10.90We have to calculate the solubility of .
Q: 1. When 2-chloropropane treated with NaOH and 1-chloropropane treated with NaOH separately produce…
A: In this question we have to complete the given reactions with proper mechanism.In next part we have…
Q: An aqueous sodium acetate, NaC,H,O,, solution is made by dissolving 0.385 mol NaC,H,O, in 0.665 kg…
A: Provided; moles of NaC2H3O2 i.e. solute 0.385 moles & mass of water i.e. solvent 0.665 Kg(say).…
Q: Compound A Compound F HCI CH3OH H₂SO4 Compound B CH3 CH₂ CH₂ Cl Compound E NaOH K₂Cr₂O7 H₂SO4…
A: To forecast the results of electrophilic additions to asymmetrical alkenes or alkynes, organic…
Q: Perform the steps above to balance the redox reaction (shown again below). Fill in each blank with…
A:
Q: ) determine the reactions's rate law according
A: To write the rate law expression ypu need to 1st write a overall reaction.
Q: Synthesis. Design a synthetic route to perform the transformations shown below. You may use any…
A: The given reaction involves the addition of carbonyl group (ester) to the aromatic compound. This…
Q: draw the best lewis structure for the free radical NO2- what is the formal charge on the N?
A: It is essential to remember that Lewis structures are simplified representations used to depict the…
Q: Draw an energy level diagram for bromine
A: Here we have to draw energy level diagram for bromine:Energy level diagram:An energy level diagram…
Q: What mass of Ca(NO3)2 is required to prepare a solution containing 0.595 mol of Ca(NO3)2?
A: For determination of the mass of Ca(NO3)2 needed for a solution with 0.595 moles of Ca(NO3)2, we…
Q: When 3.49 g of a nonelectrolyte solute is dissolved in water to make 935 mL of solution at 23 °C,…
A: mass of nonelectrolyte solute = 3.49 gVolume of the solution = 935 mL = 0.935 LOsmotic pressure (π)…
Q: 13. Which one of the following structures DOES NOT represent an aromatic compound? A N B C HN D E
A: In this question we have given some organic compounds and we have to explain that which compound is…
Q: a. As b. Cr c. Br d. Sr Write the following full and noble gas electron configurations:
A: To solve this problem we have writte the full and Nobel gas electronic configuration.
Q: Single Superphosphate (SSP) Phosphate Rock 1. Sulfuric Acid 2. Removal of Gypsum 3. Concentration +…
A: (a)The ground phosphate rock is mixed with sulfuric acid (H2SO4) in a reactor or mixer to form SSP.…
Q: A formic acid/sodium formate buffer solution can be prepared with 0.012 M each of formic acid…
A: Buffer solutions are the solutions consisting of a mixture of a weak acid and its conjugate base or…
Q: Using the thermodynamic information in the ALEKS Data tab, calculate the standard reaction entropy…
A: Using standard entropy values we can calculate the change entropy for the given reaction.
Q: Determine the solution for the following equation: (8x-8)^((3)/(2))=64 ?
A: To solve the given equation, we start by simplifying it. We rewrite 64 as 43 since 4 raised to…
Q: what is bromates structure with minimized formal charges
A: Answer :---- Bromate ion (BrO3-) has a central bromine atom bonded to three oxygen atoms. Here, in…
Q: Draw significant resonance structures for the following compound: Step 1 First, add curved arrow(s)…
A: Answer :---- Resonance structures, also known as resonance forms or canonical forms, are different…
Q: Using the thermodynamic information in the ALEKS Data tab, calculate the standard reaction entropy…
A: The entropy of a reaction (S) is the difference between the sum of the entropies of the products and…
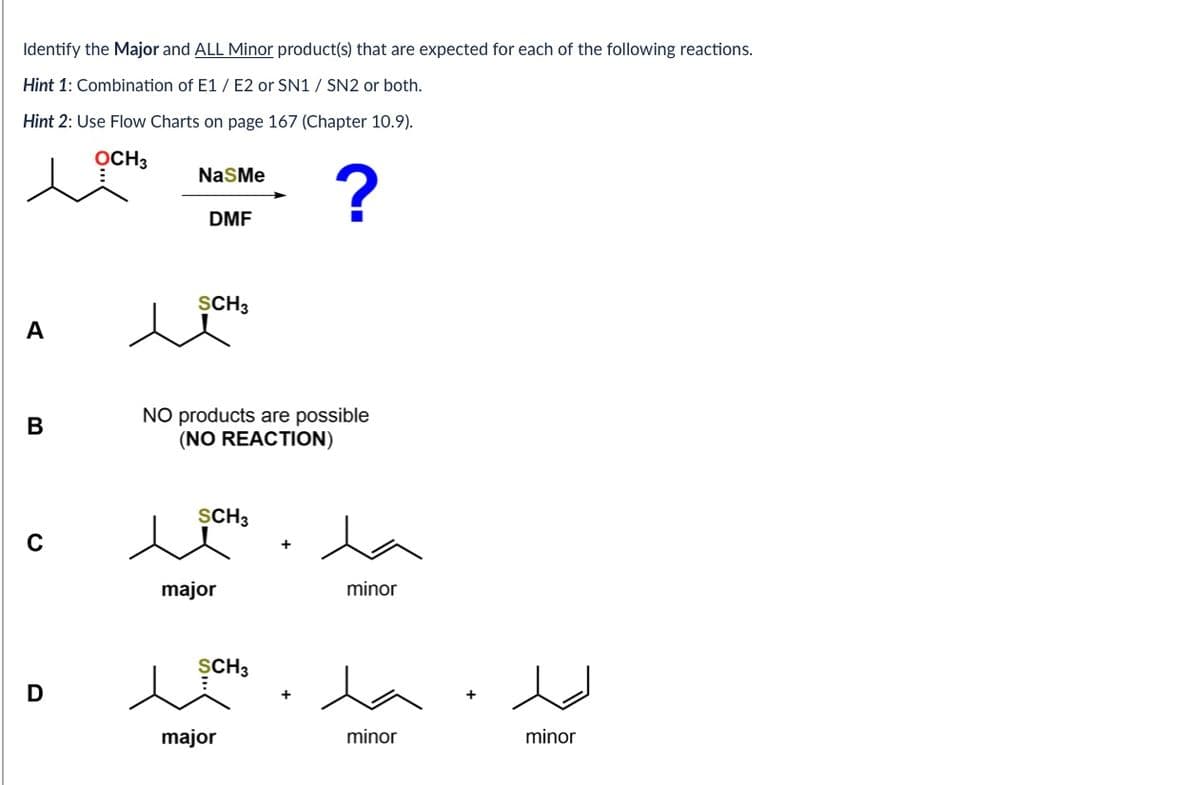
Step by step
Solved in 3 steps with 2 images

- What major product is expected from the reaction shown in Image 18?a. What are the products of following reactions?b. Write the reaction mechanism for each using the right arrows and define it as Sn2, Sn1, E2 or E1.c. Explain why do you choose that product and mechanism or in case something else happens explain why.a. What are the products of following reaction?b. Write the reaction mechanism for each using the right arrows and define it as Sn2, Sn1, E2 or E1.c. Explain why do you choose that product and mechanism or in case something else happens explainwhy.
- The synthesis above can be performed with some combination of the reagents listed below. Give the necessary reagents in the correct order. Be sure to consider stereochemistry and regiochemistry.A. NaOHB. mCPBAC. OsO4, tBuOOHD. NaHE. H2, LindlarF. CH3CH2CH2BrG. CH3CH2ClH. NaNH2I. H3O+J. Na, NH3(l)For which reaction mechanisms—SN1, SN2, E1, or E2—of thefollowing statement true? A statement may be true for one or moremechanisms. Tertiary (3°) alkyl halides react faster than 2° or 1° alkyl halides.Which reactions will proceed faster in a polar, aprotic solvent? (CH3)2CHCH2I+Na+N3−(CH3)2CHCH2I+Na+N3− (CH3)3CBr+HSCH2CH3(CH3)3CBr+HSCH2CH3 CH3CH2CH2Br+Na+CN−CH3CH2CH2Br+Na+CN− (CH3)2CHOSO2CH3+HOCH(CH3)2
- The question is: "Draw the curved arrow mechanism for the reaction between (2S,3S)-3-methylhexan-2-ol and PCl3. Note the specific instructions for each box. Include nonzero formal charges and lone pairs of electrons on all appropriate atoms" I attached screenshots of the picture of the atoms below. What type of reaction (SN1, E1, SN2, or E2) would the reaction be, and how would these molecules interact? The question asks for multiple steps so I am guessing it is either SN1 or E1, but what is the reaction mechanism?Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?What is the complete mechanism using curved arrow formalism of the two products shown below? Explain why one is major and the other is minor product formation.
- Supply the missing reagent in the following reaction: (see attachment) A. Lindlar, H2, mCPBA B. O3 or KMnO4 C. fused KOH D. NH3 E. NaNH2For which reaction mechanisms—SN1, SN2, E1, or E2—of thefollowing statement true? A statement may be true for one or moremechanisms. The reaction rate increases with better leaving groups.Hello. Kindly help me understand if this is a possible schematic of the reaction. The specific question is in Organic Chemistry Principles and Mechanisms by Joel Karty 2nd Edition, Problem 12.11 on page 614. I believe the predicted major product should come as well as with its enantiomer hence 4 products predicted for this reaction as there is no easy rotation about the cyclic structure

