Q: 3.5 Complete the following Grignard reactions. Which molecule serves as nucleophile and which one as…
A: The above complete Grignard reactions are given in attached image.
Q: Which set of starting materials can be used to make the target molecule in one step? HO. Но Target…
A:
Q: 1. Draw the four major resonance contributor of the following molecule. Show all nucleophilic atoms…
A:
Q: ) Label each of the following using N for nucleophile and E for electrophile. OH ČI H+ Он „NH2 BH3…
A: A nucleophile is a substance which is electron rich and can donate an electron pair to an…
Q: (Rank each of the molecules from least to most nucleophilic with 1 being the least four being the…
A: In chemistry, a nucleophile is a chemical species that forms bonds with electrophiles by donating an…
Q: 1. N2OCH, H3C. o-CH3 2. H,о* cH3
A:
Q: Which one of these compounds would be the best starting material for a malonic ester synthesis? CH3…
A:
Q: 6. Which of the following alkyl halides CANNOT undergo E2 elimination? (circle your answer) Br Br Br
A:
Q: Draw the least stable resonance form for the intermediate in the following electrophilic…
A:
Q: Which compound forms an anhydride more rapidly?
A: Both of the compounds are able to form anhydride compounds. The lesser the number of atoms in a…
Q: Draw the most stable resonance form for the intermediate in the following electrophilic substitution…
A: Electrophilic aromatic substitution In an electrophilic aromatic substitution process, a Lewis acid…
Q: For the compound below choose the position of the carbon (1- target for reaction of aromatic…
A: Electrophile attack on that position which has high electron density
Q: Rank the following compounds in order of increasing reactivity in a substitution reaction with −CN…
A: The rate of reaction in which each given compound undergoes will depend only on the leaving group…
Q: 8-CH3
A: Ans
Q: Encircle all nucleophilic centers in each of the following compounds: OH (c) ZHN (b) (d)
A: Nucleophiles are the nucleus-loving species that possess a high density of electrons. Nucleophilic…
Q: Which of the following nucleophiles would add to an alpha, beta- unsaturated ketone via direct…
A: Given Reactant Alpha,beta-unsaturated ketone reagent which give Direct addition = to be determined
Q: C. NaOCH3 d. NaOH Ph
A:
Q: Match each of the following questions with the pool of products. WRITE THE ANSWER IN CAPITAL…
A: In SN2 reactions inversion of configuration takes place . In E2 reactions the product formed is…
Q: The following compounds are treated with HNO3/H2SO4. Predict the positions of electrophilic attack…
A: In nitrarion reaction, NO2+ electrophile attacks on the position of the benzene where electrons…
Q: Draw a structure for the major product in each of the following reactions DMSO OTs CI NaOCH;
A: An acyl substituted compound and an alkene will be the major products for these given reactions.
Q: Which substrate reacts with the cyanide anion ("CN) most rapidly in an SN2 reaction? O CH3CH2F O…
A: Nucleophilic substitution biomolecular reaction (SN2):The bimolecular nucleophilic substitution…
Q: When a single compound contains both a nucleophile and a leaving group, an intramolecular reaction…
A: A nucleophilic substitution reaction is defined as the reaction in which a nucleophile attacks on…
Q: Complete the curved arrow mechanism of the following double elimination reaction when…
A: In the organic reaction mechanism, the curved arrow shows the movement of the electron pair. In the…
Q: Draw the major E2 elimination products from each of the following alkyl halides.
A: For E2 elimination reaction the leaving group and the Hydrogen on the Beta-Carbon should be trans to…
Q: How does the rate of an SN1 reaction change as the alkyl group in the substrate alkyl halide changes…
A:
Q: Which molecule would be most susceptible to re-arrangement during an E1 attack, as the electrophile?…
A: Ans: 2-Bromo-3-methylhexane involves rearrangement. See below for mechanism.
Q: 1. For each of the following molecules, put a box around the nucleophilic atom(s). MgBr S H.
A:
Q: What is the major product formed when the following compound undergo an E1 reaction? CH, -CH;CH;
A: Solution An chemical action may be a variety of organic reaction throughout that two substituents ar…
Q: What will be the major product of the shown E2 reaction: CH3 H3C-C-o CH3 acetonitrile H. Br…
A: The elimination reaction E2 is a bimolecular elimination, which has only one step. A new bond is…
Q: Which of the following are most likely to behave as electrophiles? * (a) NH,+ (b) C=N- (с) Brt (d)…
A: Br2 can undergo heterolytic cleavage to form Br- and Br+ ions. Br+ is very unstable due to lack of…
Q: 6. In the conversion in the box which of the following acts as a nucleophile? excess NH3 CI `NH2 +…
A:
Q: 1. NaOCH3 H3C CH3 2. H,0+ CH3
A:
Q: (Rank the substrates in order of increasing reactivity in an SN2 reaction. (1 = least reactive, 4 =…
A: SN2 reaction proceeds via a one step mechanism in which the bond breaking and bond forming takes…
Q: What direction does the nucleophile approach the substrate in an SN2 reaction?
A: Simple and small groups under go SN2 reaction but heavily substituted cannot under go SN2 reaction.…
Q: H,0 Cat TSOH но OMe OMe MEOH cat. TSOH -H0
A: Ring opening reaction of epoxides goes through SN2 mechanism.
Q: Which of the following bases can remove a proton from a terminal alkyne in a reaction that favors…
A: The ions or molecules which have more electronegativity value than the electronegativity of C in…
Q: 1. For each of the following molecules, put a box around the nucleophilic atom(8). MgBr SH H.
A: A nucleophile is a species which is highly electron rich in nature while an electrophile is defined…
Q: Type of substrates undergo SN2 & SN1 reactions & 3 examples SN2 SN1
A: A reaction in which an atom or group of atoms is replaced by another atom or group of atoms is known…
Q: Pls help ASAP.
A: To solve this problem we have to know about the mechanism of E1 reaction.
Q: Which of the following combination has the slowest esterification reaction rate? 1. CH3CH2CH2COOH IL…
A: Correct option is II & IV
Q: Draw the product formed when the compound shown below undergoes a reaction with HCl in CH,Cl2.…
A: The given compund is an alkene. it is 2-butene. when the given compound(alkene) is reacted with HCl,…
Q: 1. NaOEt CaH12O2 OEt 2. H3O*
A: To
Q: SO,H CH2-CH II
A:
Q: OH SOCI2 pyridine NaCN
A: SN2 reaction is a bimolecular nucleophilic substitution reaction which occurs in one step. Here the…
Q: Which position is activated toward electrophilic aromatic substitution? C O A O D OE B.
A: Option 2nd (D) will be correct.
Q: Which of the following is the strongest activating group in electrophilic aromatic substitution…
A: Aromatic electrophilic substitution reaction: Benzene and other aromatic compound gives mainly…
Q: Me Me OMe 9. 10
A: We are given two eater molecules:
Q: There are three possible products for the E1 elimination of this reaction. Draw one of the major…
A:
Q: 1c. What is the nucleophile in the following reaction? Br CH;COO Na L0OCCH3 + + NaBr 1d. What is the…
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Match the questions with the correct descriptions below: Sn2, E2 E1cb Sn1, E1 (anti) E2, E1cb Sn1, E1 2 steps poor leaving group 2 carbons removed from a carbonyl group carbanion intermediate 3°>allylic, benzylic>2°>1°>CH₃ carbocation intermediate Ist order reaction (r=k[RX]) H-X anti no intermediate 2nd order reaction (r=k[RX][Nu]) polar aprotic solventConsider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:Which of the following bases can remove a proton from a terminal alkyne in a reaction that favors products? CH3O− NH3 CH3C-H2 H2=C-H F-
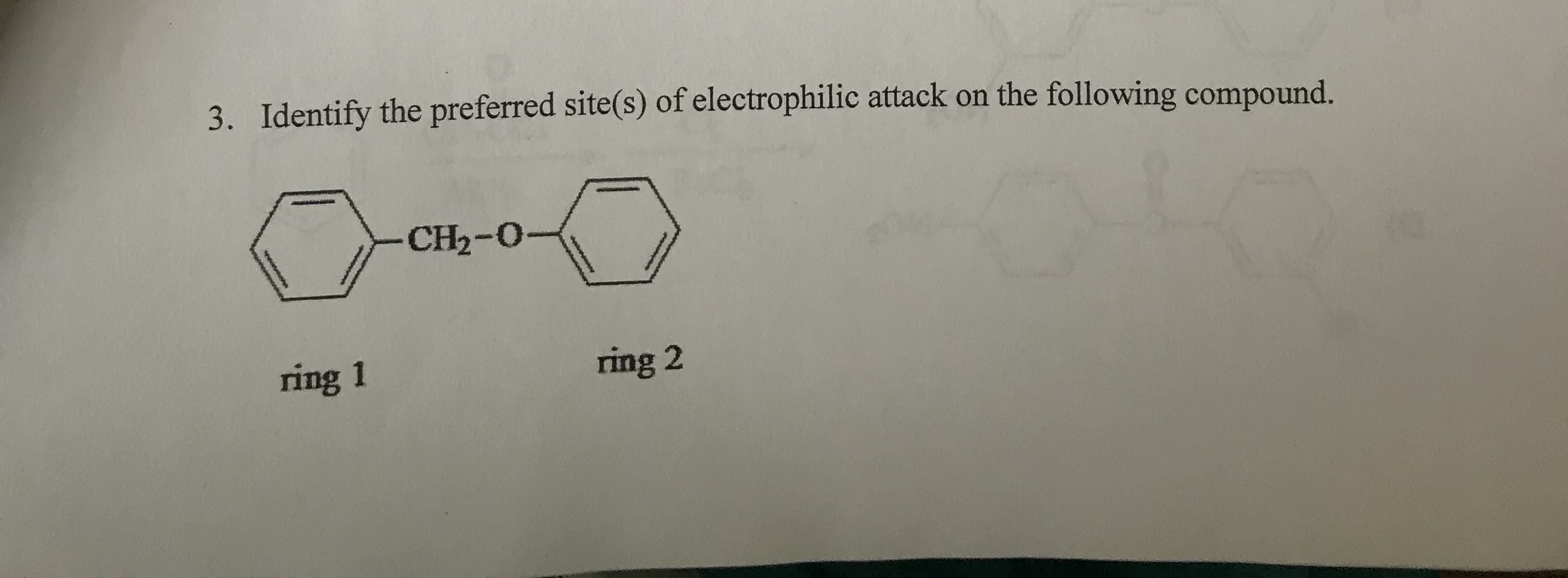
- Identify the stronger nucleophile in the following pair ? CH3CO2−, CH3CH2O−Identify the preferred site(s) of electrophilic attack on the compound shown in Image 5. A. ortho/para positions on ring 1 B. meta position on ring 1 C. ortho/para positions on ring 2 D. ortho/para positions on ring 2How does the rate of an SN1 reaction change as the alkyl group in the substrate alkyl halide changes from CH3 1° 2° 3°?
- Identify the stronger nucleophile in each pair. a. NH3,−NH2 b. CH3NH2, CH3OH c. CH3CO2−, CH3CH2O−Which of the following bases can remove a proton from a terminal alkyne in a reaction that favors products? CH3O− NH3 CH3CH2 H2C=CH F−10. Draw the most stable resonance form for the intermediate in the following electrophilic substitution reaction.



