Q: A system of N particles with K number of holonomic constraints, the degree of freedom? O N-K O 3N -…
A: For a particle there are 3 degrees of freedom . And holonomic constraints are relations between the…
Q: Isoamyl acetate 100 95 90 85 80 75 70 65 60 55 50 45 40 4000 3500 3000 2500 2000 1500 1000 500 Wave…
A:
Q: in a 1.00-cm cell. What is the molar absorptivity at 266 nm? 3.55 x 104 M-1-cm-1 6.54x 104 M-1.cm-1…
A: Given that: %Transmittance = 15.1% Conc.(C) = 2.31×10-5 M Length(l) = 1.00cm
Q: Calculate the wavenumber of the first overtone transition with v = 2 ← O in nitric oxide. NO. given…
A: Given information:
Q: I need the theoretical molar extinction coefficient of paranitrophenol in mol^-1cm^-1 (using moles…
A: Molar extinction coefficient is an intrinsic property of a compound which doesn't depend on…
Q: Distinguish between voltammetry and amperometry
A: 1) Voltammetry is an analytical technique in which variable potential is applied to the working…
Q: The wavenumber of the incident radiation in a Ramanspectrometer is 20 623 em·'. What is the…
A: The wavenumber of the incident radiation in a Raman spectrometer is 20623 cm-1. What is the…
Q: Parameters to use for the problems that follow: K = 1.38 × 10-23 J/K q = 1.6 × 10-¹⁹ coulombs For…
A: Solutions- Given data- Nd = 4 x 1015 cm-3 Ni = 1 x 1010 cm-3 T = 300K
Q: Let the retention factor of species A be 0.5, while the retention factor of species B be 0.3. If…
A: Given Rf = Retention factor Rf(A) = 0.5 Rf(B) = 0.3 Distance travelled by solute A = 1.0 cm
Q: Calculate the temperature at which qvib = 2.5 for nitric oxide. The vibrational wavenumber is = 1876…
A: answer is given below:
Q: Given some molecule given their vapor pressure of a number of liquids. in which who have the highest…
A: Intermolecular forces are the forces present in between the molecules. Stronger is the…
Q: An analyst obtained the following data for the percent compound Z in triplicates (n-3) of an…
A: Solution Given that Z in triplicattes (n) = 3 Hence option( D) is…
Q: For 1H35Cl molecule, (N1/N0) ratio at 300 K is 6.091 x 10-2. Calculate the wavenumber ( in cm-1) for…
A: For 1H35Cl molecule, (N1/N0) ratio at 300 K is 6.091 x 10-2. The wave number is 582.741Cm-1
Q: The following data on intrinsic viscosities and GPC peak elution volumes at 25°C fo= standard…
A: Mw×10-3 log Mw Ve 867 2.938019 149 411 2.613842 157 173 2.238046 177…
Q: Construct the expression for Ksp for solid Ba3(PO)2 in aqueous solution.
A:
Q: C: Coordination Number Cu2+ Mass Cu(NO,)2 -3 H20 2.3366 grams Volume D.I. Water 57.0 mL Trial…
A: Given:
Q: Transmittance (%) 100% 75% 50% 25% 0% 4000 3500 3000 مل ОН 2500 2000 Spectrum 3B 1750 سمه 1500…
A: The bonds of molecules absorb the infrared radiation and vibrate. IR spectroscopy detects the…
Q: Construct the expression for Ksp for solid Ba3(PO4)2 in aqueous solution. Ba3(PO.)2(s) = 3 Ba"(aq) +…
A: The expression for Ksp for solid Ba3(PO4)2 in aqueous solution has to be given.
Q: The wavenumber of the incident radiation in a Raman spectrometer is 20 487 cm−1. What is the…
A: Given the rotational transition of 14N2 molecule, Wavenumber of incident radiation in Raman…
Q: Calculate the wavenumber (cm^-1) and excitation energy (kj/mol) for each of the following max…
A: Electromagnetic spectrum involves different radiations from the lowest to the highest frequency…
Q: Compute the relative population of states of an electronic spin in magnetic field of 0.3T at 300K…
A:
Q: P14C.2 The surface tensions of a series of aqueous solutions of a surfactant A were measured at…
A: The surfactant is A Surface tensions of a series of aqueous solutions of this surfactant were…
Q: Calculate the vibrational energy of the v = 2 state of 1H127 I relative to the ground state,…
A: The vibrational energy of the v = 2 state of 1H127I relative to the ground state is to be…
Q: The resolution product of Ag2S03 is 1.5 x 10-14. Ag2SO3 (k) + 2e" + 2 Ag (k) + S032- Calculate E0…
A:
Q: IR isoamyl alcohol 100 95 90 85 80 75 70 65 60 55 50 45 40 4000 3500 3000 2500 2000 1500 1000 500…
A:
Q: Estimate the centrifugal distortion constant for 79Br81Br, for which ᷉ B = 0.0809 cm−1 and ᷉v =…
A: A molecule is microwave active if and only if they have a permanent dipole moment, and hence they…
Q: The resolution product of Ag2SO3 is 1.5 x 10-14. Calculate E0 for Ag2SO3(s) + 2e- ↔ 2 Ag(s) +…
A: The answer is explained in the image below.
Q: Calculate the p-functions for each ion in a solution that is i. 0.020M in KCI ii. 2.7 x 103М in…
A: The p function is calculated by taking the negative logarithm of the concentration of ion present in…
Q: If you are using a NORMAL phase HPLC, predict the order of elution of 1. n-hexane, n-hexanol and…
A:
Q: Provided the following information determine ΔHformation of C2H6(g). C(graphite) + O2(g) ⟶…
A:
Q: The resolution product of Ag2SO3 is 1.5 x 10-14. Calculate E0 for Ag2SO3 (k) + 2e- ↔ 2 Ag (k) +…
A: Solubility product ksp= 1.5 ×10-14 E°Ag+/Ag = 0.799v Calculate the E for Ag2SO3 calculate the…
Q: 0.8- 0.6- 0.4 0.2 3000 2000 Wavenumber (cm-1) 1000 Relative Transmittance
A: IR spectroscopy can predict only the functional groups. Here significant IR peaks are at 1) 2950…
Q: The resolution product of Ag2SO3 is 1.5 x 10-14. Calculate E0 for Ag2SO3 (k) + 2e- ↔ 2 Ag(k) +…
A:
Q: Bb MYJCC Blackboard Learn WebAdvisor for Students JCC Self Service Bb UTF-8 Syllabus_CHM%20151%2 X A…
A: As per regulations, only three sub-parts can be answered.
Q: 1. Calculate the thermal average microscopic absorption cross section at 200 °C for: a) b) c)…
A: The average cross-section is calculated as follows: σ=∫σEϕEdE∫ϕEdE Here,…
Q: Does the data set of 155.8, 158.4, 194.9, 164.8, 154.5, 184.3, 114.8 and 157.8 ppm contain…
A: Outlier is the point which is farthest from the original path or line.
Q: For 1H35Cl molecule, (N1/N0) ratio at 300 K is 6.091 x 10^-2. Calculate the wavenumber ( in cm^-1 )…
A: Answer is explained below. Given that, for 1H35Cl molecule (N1/N0) = 6.091×10-2 at 300K. Now, we…
Q: When are isosbestic points observed and why?
A: Isosbestic points refer to point at certain specific wavelength when absorbance spectrum of two pure…
Q: 0.8- 0.6 0.4 0.2- 3000 2000 Wavenumber (cm-1) 1000 TRANSMITTANCE
A:
Q: 0.2- 3000 2000 Wavenumber (cm-1) 1000 200 180 160 140 120 100 80 60 40 20 CDS-3-414 ppm HO, NH2 B. D
A: B option is correct
Q: Construct the expression for Ksp for solid Bas(PO4)2 in aqueous solution. Ba:(PO:):(s) = 3 Ba2"(aq)…
A: • The dissociation of the sparingly soluble salt Ba3(PO4)2 is, Ba3(PO4)2(s) ⇌ 3 Ba2+(aq) + 2…
Q: ए नैमट म्रभ (लाCOTकनीना ड ड.25K10-4M the abseved the cett potentiat at जीाट विडव्ण्ववं जेकए 298 k…
A: The step wise solution is as follows -
Q: INFRARED SPECTRUM 0.8 0.6 OH 0.4 OH 0.2 3000 2000 1000 Wavenumber (cm-1) TRANSMITTANCE
A: In first IR, the correct answer is D. A strong peak observed around 1680-1690 cm-1 is attributed…
Q: P1B.1 A rotating slotted-disc apparatus consists of five coaxial 5.0 cm diameter discs separated by…
A: Rotating disk apparatus (RDA) is an instrument primarily containing a turning plate and an…
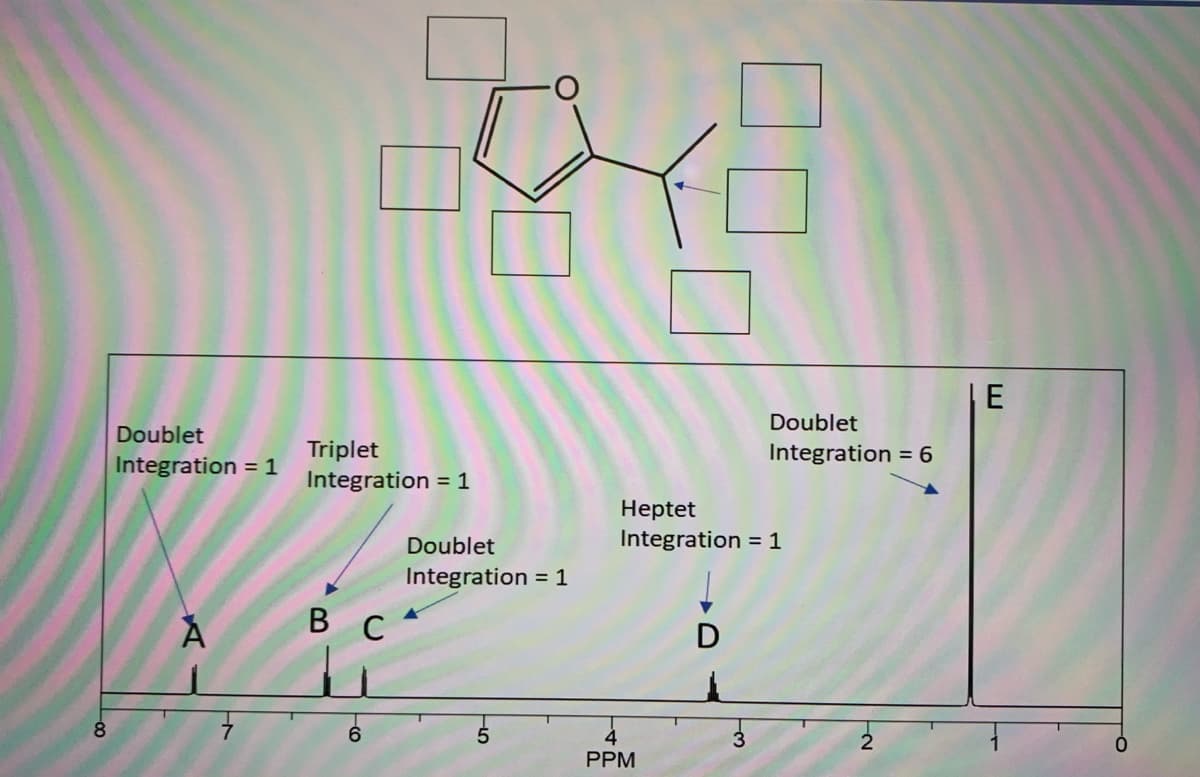
Q: Use the 1H NMR and IR spectra given below to identify compound A, having molecular formula C4H8O2.
A: Number of signals in 1H NMR: Tells us how many sets of protons with different electronic environment…
Q: For a distribution Mean, Median and Mode are 23,24 and 25.5 respectively, then it is most likely…
A: For a distribution Mean, Median and Mode are 23,24 and 25.5 respectively, then it is most likely…
Q: Isoamyl acetate 100 90 80 70 60 50 40 30 20 10 4000 3500 3000 2500 2000 1500 1000 500 wave…
A:
Q: Intensity (a.u.) 4000 3352 cm¹ 3500 3000 UNKNOWN B 1710 cm¹ 1668 c cm¹ 2500 Wavenumber (cm¹¹) 2000…
A: Ir spectroscopy helps in the identifiaction of organic compounds.It helps in determining functional…
Step by step
Solved in 2 steps with 2 images

- Calculate the ΔHsoln and ΔSsoln for the dissolution of solid KHP from the trendline equation of your scatter graph. Include units. Reffer to equation 4. R is the gas constant 8.314J mol-1 K-1 The trendline equation -5623*x + 17.7 Equation 4 Ln Ksp = - ΔHsoln/R (1/T) + ΔSsoln/ Rstate 2 advantages of transitional theory over collision theoryPolymer PXX has a glass transition temperature of 49°C What inter-molecular interactions are present and what is the consequent effect upon molecular motion?
- The difference in a chemical potential of a particular substance between two regions of a system is -8.3 kJ/mol. By how much does the Gibbs energy change when 0.15 mmol of that substance is transferred from one region to the other?Carbonic anhydrase of erythrocytes (Mr 30,000) has one of the highest turnover numbers known. It catalyzes the reversible hydration of CO2:This is an important process in the transport of CO2 from the tissues to the lungs. If 10.0 μg of pure carbonic anhydrase catalyzes the hydration of 0.30 g of CO2 in 1 min at 37 °C at Vmax , what is the turnover number (kcat) of carbonic anhydrase (in units of min −1 )?The change in the molar volume accompanying fusion of solid CO2 is −1.6 cm3 mol−1. Determine the change in the molar Gibbs energy of fusion when the pressure is increased from 1 bar to 1000 bar.
- Formula for work done in isobaric process(according to chemistry thermodynamics)An effusion cell has a circular hole of diameter 2.50 mm. If the molar mass of the solid in the cell is 260 g mol-1and its vapour pressure is 0.853 Pa at 400K, by how much will the mass of the solid decrease in a period of 2.00 hours?13.3p. If you allow one pound of carbon dioxide to sublime (change from CO2(s) to CO2(g)) at room conditions of roughly 1atm and 25C, what volume would it occupy?
- Calculate the change in S when one mole of water is heated from 263 to 283 K given the molar capacities inJ.K-1 , Cp(ice) = 2.09 + 0.126T, Cp(water)=75.3, and change in Hm=6000Jmol-1Identify the systems for which it is essential to include a factor of 1/N! on going from Q to q : (i) a sample of carbon dioxide gas, (ii) a sample of graphite, (iii) a sample of diamond, (iv) ice.The pressure dependence of G is quite different for gases and condensed phases. Calculate ΔGm for the process (C,solid,graphite,1bar,298.15K)→(C,solid,graphite,300.bar,298.15K) The density for graphite is 2250 kg⋅m−3 Calculate ΔGm for the process (He,g,1bar,298.15K)→(He,g,300.bar,298.15K)

