If there is a table such as this one given with Ksp values, does a higher Ksp mean that the kidney stones break easier since they are more soluble? Or is it the opposite?
If there is a table such as this one given with Ksp values, does a higher Ksp mean that the kidney stones break easier since they are more soluble? Or is it the opposite?
Chapter79: Solubility
Section: Chapter Questions
Problem 1P
Related questions
Question
If there is a table such as this one given with Ksp values, does a higher Ksp mean that the kidney stones break easier since they are more soluble? Or is it the opposite?
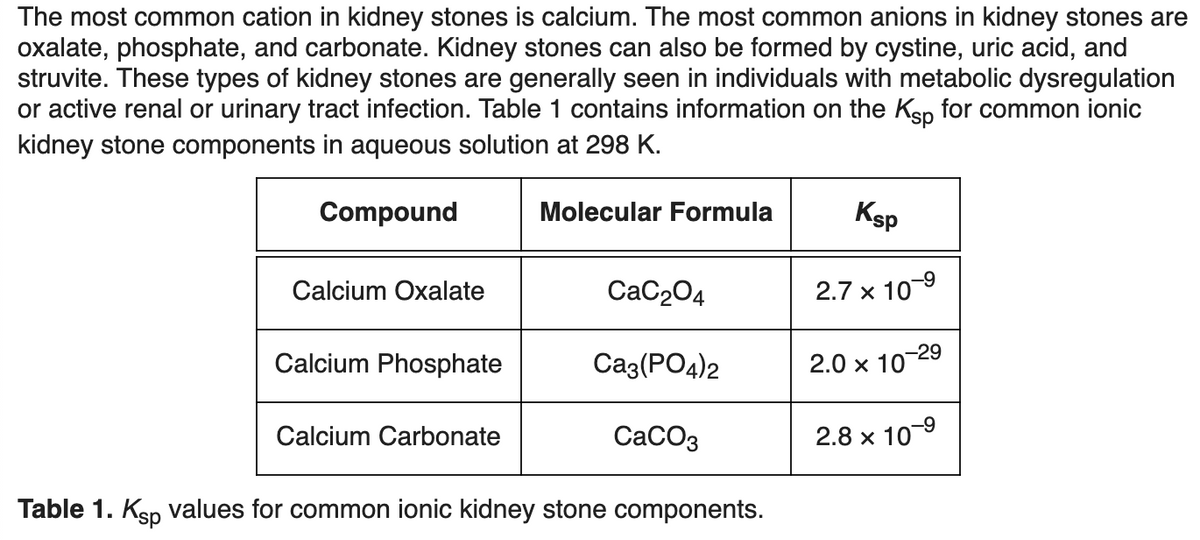
Transcribed Image Text:The most common cation in kidney stones is calcium. The most common anions in kidney stones are
oxalate, phosphate, and carbonate. Kidney stones can also be formed by cystine, uric acid, and
struvite. These types of kidney stones are generally seen in individuals with metabolic dysregulation
or active renal or urinary tract infection. Table 1 contains information on the Kåp for common ionic
kidney stone components in aqueous solution at 298 K.
Compound
Calcium Oxalate
Calcium Phosphate
Calcium Carbonate
Molecular Formula
CaC₂04
Ca3(PO4)2
CaCO3
Table 1. Ksp values for common ionic kidney stone components.
Ksp
2.7 x 10-⁹
-29
2.0 x 10
2.8 x 10
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT