In chemical reactions, heat is converted into chemical energy (the potential energy stored in chemical bonds) or vice versa. Bond energy is the energy required to break one mole of the bond in the gas phase. Since it takes energy to break a bond, bond energies are always positive. Conversely, energy is released when bonds are formed. Thus, the enthalpy change for a reaction can be approximated from Part A Calculate the bond energy per mole for breaking all the bonds in methane, CH4. ΔΗ Σ(ΔΗ breaking) + Σ(ΔΗ forming) Express your answer to four significant figures and include the appropriate units. where H represents bond energies for the breaking (positive bond energy) or forming (negative bond energy) of a bond and Hrxn represents the overall enthalpy for the • View Available Hint(s) reaction. Use the table to answer questions about bond energies. HẢ Bond energy Bond (kJ/mol ) Value Units AHCH, = C-C 347 C=C 611 Submit Сн 414 C-O 360 Part B C=OinCO2 799 O-0 142 Calculate the bond energy per mole for breaking all the bonds of oxygen, O2? 0=0 498 Express your answer to three significant figures and include the appropriate units. но 464 • View Available Hint(s) HA Value Units ΔΗo, - Submit
In chemical reactions, heat is converted into chemical energy (the potential energy stored in chemical bonds) or vice versa. Bond energy is the energy required to break one mole of the bond in the gas phase. Since it takes energy to break a bond, bond energies are always positive. Conversely, energy is released when bonds are formed. Thus, the enthalpy change for a reaction can be approximated from Part A Calculate the bond energy per mole for breaking all the bonds in methane, CH4. ΔΗ Σ(ΔΗ breaking) + Σ(ΔΗ forming) Express your answer to four significant figures and include the appropriate units. where H represents bond energies for the breaking (positive bond energy) or forming (negative bond energy) of a bond and Hrxn represents the overall enthalpy for the • View Available Hint(s) reaction. Use the table to answer questions about bond energies. HẢ Bond energy Bond (kJ/mol ) Value Units AHCH, = C-C 347 C=C 611 Submit Сн 414 C-O 360 Part B C=OinCO2 799 O-0 142 Calculate the bond energy per mole for breaking all the bonds of oxygen, O2? 0=0 498 Express your answer to three significant figures and include the appropriate units. но 464 • View Available Hint(s) HA Value Units ΔΗo, - Submit
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter6: Covalent Bonding
Section: Chapter Questions
Problem 98QRT
Related questions
Question
100%
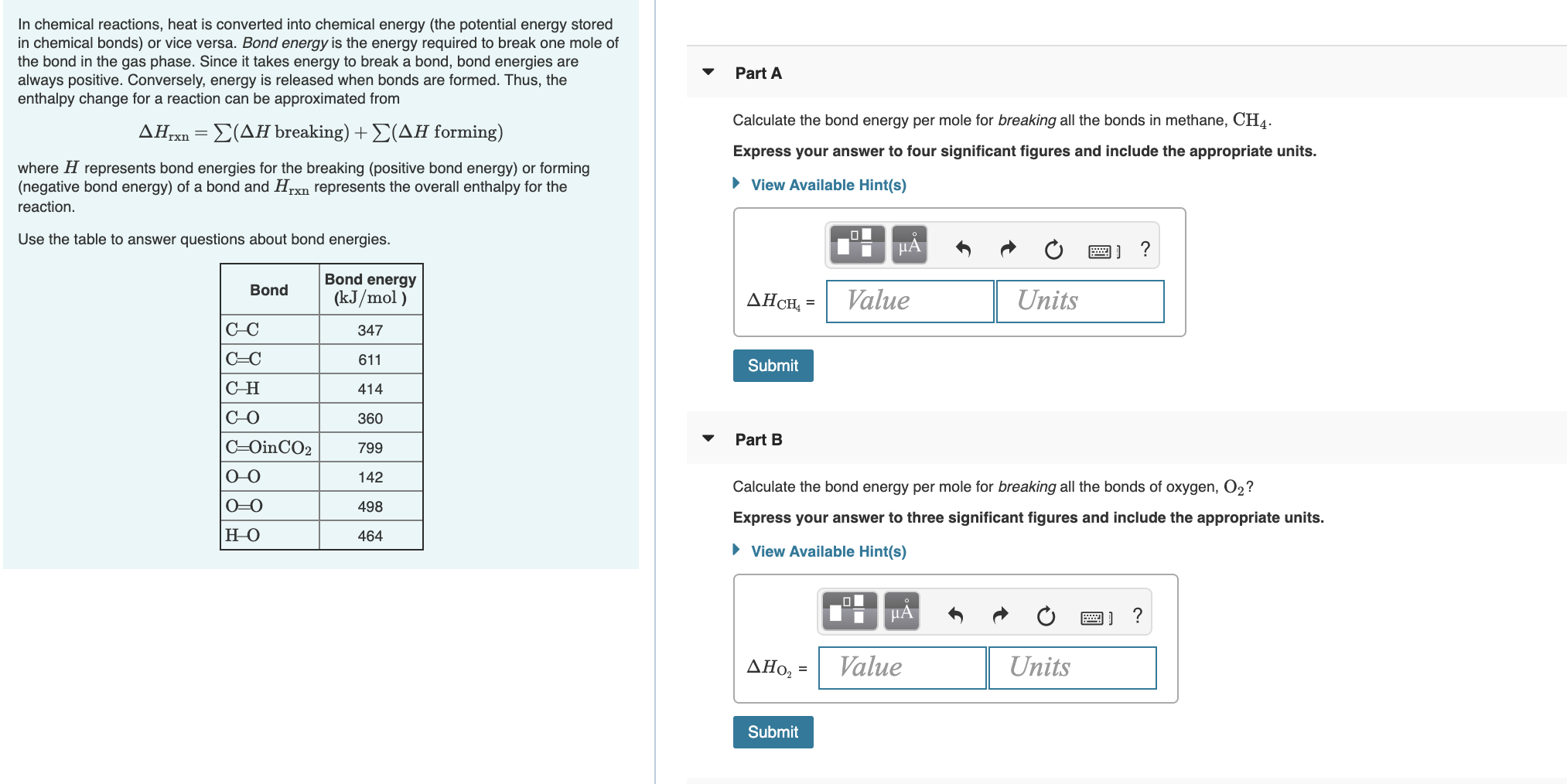
Transcribed Image Text:In chemical reactions, heat is converted into chemical energy (the potential energy stored
in chemical bonds) or vice versa. Bond energy is the energy required to break one mole of
the bond in the gas phase. Since it takes energy to break a bond, bond energies are
always positive. Conversely, energy is released when bonds are formed. Thus, the
enthalpy change for a reaction can be approximated from
Part A
Calculate the bond energy per mole for breaking all the bonds in methane, CH4.
ΔΗ
Σ(ΔΗ breaking) + Σ(ΔΗ forming)
Express your answer to four significant figures and include the appropriate units.
where H represents bond energies for the breaking (positive bond energy) or forming
(negative bond energy) of a bond and Hrxn represents the overall enthalpy for the
• View Available Hint(s)
reaction.
Use the table to answer questions about bond energies.
HẢ
Bond energy
Bond
(kJ/mol )
Value
Units
AHCH, =
C-C
347
C=C
611
Submit
Сн
414
C-O
360
Part B
C=OinCO2
799
O-0
142
Calculate the bond energy per mole for breaking all the bonds of oxygen, O2?
0=0
498
Express your answer to three significant figures and include the appropriate units.
но
464
• View Available Hint(s)
HA
Value
Units
ΔΗo, -
Submit
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning