In some aquatic ecosystems, nitrate (NO3 ) is converted to nitrite (NO2), which then decomposes to nitrogen and water. As an example of this second reaction, consider the decomposition of ammonium nitrite: NH NO2(ag) N2(g)+2H20(1) 1st attempt See Hint See Periodic Table What would be the change in pressure in a sealed 10.0 L vessel due to the formation of N2 gas when the ammonium nitrite in 1.00 L of 0.600 M NH4NO2 decomposes at 25.0°C? atm
In some aquatic ecosystems, nitrate (NO3 ) is converted to nitrite (NO2), which then decomposes to nitrogen and water. As an example of this second reaction, consider the decomposition of ammonium nitrite: NH NO2(ag) N2(g)+2H20(1) 1st attempt See Hint See Periodic Table What would be the change in pressure in a sealed 10.0 L vessel due to the formation of N2 gas when the ammonium nitrite in 1.00 L of 0.600 M NH4NO2 decomposes at 25.0°C? atm
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter5: Gases
Section: Chapter Questions
Problem 5.60PAE: 60 Automakers are always investigating reactions for the generation of gas to inflate air bags, in...
Related questions
Question
really struggling with this.
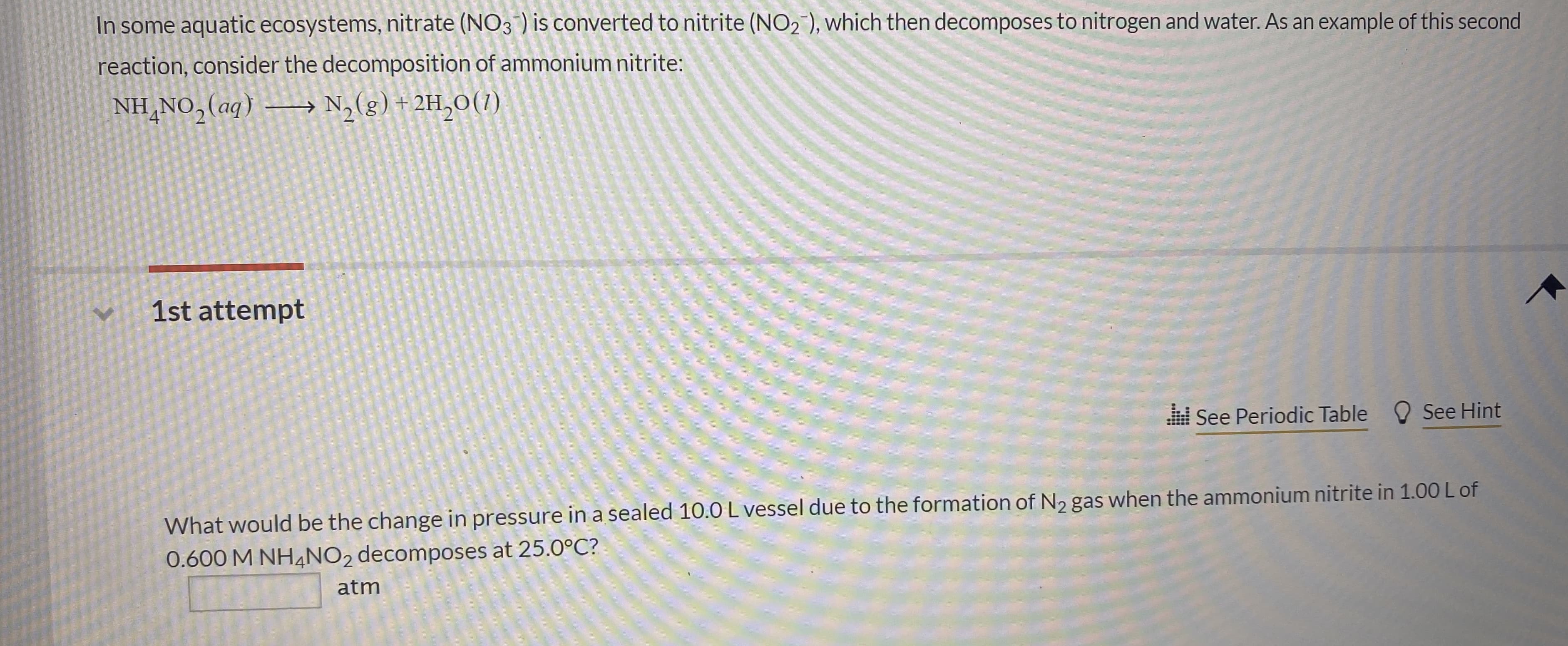
Transcribed Image Text:In some aquatic ecosystems, nitrate (NO3 ) is converted to nitrite (NO2), which then decomposes to nitrogen and water. As an example of this second
reaction, consider the decomposition of ammonium nitrite:
NH NO2(ag) N2(g)+2H20(1)
1st attempt
See Hint
See Periodic Table
What would be the change in pressure in a sealed 10.0 L vessel due to the formation of N2 gas when the ammonium nitrite in 1.00 L of
0.600 M NH4NO2 decomposes at 25.0°C?
atm
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning