Q: Consider the following two reactions and the corresponding enthalpy changes: CH4(g) +2 O2(g)→…
A:
Q: construct a balanced reaction for the formation of Hg2Cl2(s) from its elements in their standard…
A: Answer: Standard state of an element means its physical state in which it exists at 250C temperature…
Q: 1h) What will be the product of the following reaction? -Br + (HO)₂B Pd(OAc), Degassed H₂O/Acetone
A:
Q: 1i) What will be the product of the following reaction? J Br + (HO)₂B- Pd(OAc)₂ Degassed H₂O/Acetone
A: An incomplete reaction
Q: Phosphorus and bromine react to form phosphorus tribromide, like this: P4(g)+6 Br₂(g) →4 PBr3(9)…
A: The given equilibrium reaction is as follows: P4(g) +6Br2(g) ⇌ 4PBr3(g) The pressure equilibrium…
Q: Be sure to answer all parts. Determine the concentration of an aqueous solution that has an osmotic…
A:
Q: Convert 28.01 moles of X5Z5 into grams of the same compound. (Assume that the molar mass of X is…
A:
Q: Write the structures of products of the following reactions for the synthesis of saccharin
A: Saccharin is an artificial sweetener. The IUPAC name is 1,2-benzisothiazolin-3-one-1,1-dioxide.…
Q: Give the complete IUPAC name for each of the following compounds.
A: Given : structure of molecules
Q: Question: How can this IR graph of the crude product be an evadience that proof it is pure and the…
A: We need to provide reasons how the IR of crude product is similar to the product obtained from the…
Q: Macmillan Learning If the K, of a monoprotic weak acid is 3.3 x 10-6, what is the pH of a 0.41 M…
A:
Q: Write a detailed mechanism for the following reaction. Draw the structures of all the expected…
A: In this question, we will give reaction mechanism and also find out the products A, B and C In…
Q: A chemical engineer is studying the following reaction: 2 H₂S(g)+30₂(g) → 2 SO₂(g)+2H₂O(g) - At the…
A: The direction in which the reaction proceeds to reach equilibrium can be predicted by determining…
Q: What mass of Na2SO4 is necessary to fully react with 25 L of .01M of PbCl2 solution?…
A:
Q: Which of the followinf salts could be added to the PbCl2 solution to decrease Pb2+ concebtration,…
A: Salts are ionic compounds formed by the chemical reaction between an acid and a base. They are…
Q: A certain reaction is first order in N₂ and second order in H₂. Use this information to complete the…
A:
Q: In this section, describe the compound, ethanol. You must include the following information:…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: Calculate the molality of a 0.845 M aqueous solution of cadmium bromide with a density, p= 1.029…
A:
Q: What will be the product of the following reaction? -Br + (HO)₂B- Pd(OAc)₂ Degassed H₂O/Acetone
A: -> Suzuki reaction is an organic reaction classified as a cross coupling reaction when the…
Q: Where did you get the 1.6*10^-19 and the 9.1*10^-31?
A: This question is related to atoms. There are mainly three subparticles in an atom- Electron Proton…
Q: How many atoms of oxygen are in25 gofSO2? a)2.3×1022 b)6.8×1023 c)1.4×1023 d)4.6×1023…
A: The number of atoms of oxygen present in 25 g of SO2 are The number of moles of carbon present in…
Q: Beryllium-10 changes to boron-10 . write a balanced nuclear equation and identify the radiation…
A: Given -> Beryllium-10 ----> Boron-10
Q: Enter your answer in the provided box. Determine the percent sulfuric acid by mass of a 1.21 m…
A:
Q: The equilibrium constant for the reaction N2(g)+3H2(g)←→2NH3(g)N2(�)+3H2(�)←→2NH3(�) is…
A:
Q: Draw the correct product for the reaction. (If there is no reaction, draw the starting material.) OH…
A:
Q: Calculate the molarity of a 1.03 m aqueous solution of tin(IV) nitrate with a density p= 1.082 g/mL.…
A:
Q: The resonance hybrid for the commercial dye, Basic red 1, will show a__ charge on the nitrogen atom…
A: Formal charge: The formal charge (FC) is the charge assigned to an atom in a molecule, assuming that…
Q: 4. Write the formulas of the following compounds: (a) phosphorus triiodide (d) diphosphorus…
A: Answer-4 When an atom forms more than one type of compound with the same other element in than for 1…
Q: Draw the sulfur-containing product of the oxidation reaction between two 2-methyl-1-propanethiol…
A: In this question, we will draw the sulfur containing product by the oxidation of thiol compound.
Q: What is the correct IUPAC name of the following compound? H H CH₂CH3 CH₂ Multiple Choice…
A: Introduction IUPAC nomenclature is a system of naming chemical compounds and identifying the…
Q: What is the identity of the uknown metal?
A: Compare calculated the density of unknown metal with the given densities of metals. given: density…
Q: A C 1.2 % NaCl 280 mOsm/L B D 5% Glucose 2% Glucose Assuming equal volume in all the sections, what…
A: Here it is given that the volume of all the sections are same. So A, B, C and D have se volume. We…
Q: you have a solid mixture that contains lithium bromide and barium carbonate. use the handbook of…
A: There are several methods to separate chemical compounds, including: Distillation: used to separate…
Q: Using the following thermodynamic data, calculate the lattice enthalpy of lithium oxide: Li(g) →…
A: It is based on the concept of lattice energy Here we are required to find the lattice energy of…
Q: A gas contracts to half its initial volume while releasing heat to the surroundings. What can be…
A: Given : heat is releasing to the surrounding And gas contract
Q: Select all that apply. Sight down the C-2-C-3 bond, and choose the Newman projection formulas for…
A: Answer:- This question is answered by using the simple concept of stability of conformer which…
Q: A 0th order chemical reaction was determined to have a reaction constant k = 0.00123 M/s. If the…
A:
Q: 4) Use data for AG°f to calculate equilibrium constants for each reaction at 25C: J ·R = (31 Mal R…
A:
Q: In the laboratory, a general chemistry student measured the pH of a 0.392 M aqueous solution of…
A:
Q: i Add the missing curved arrow notation.
A: Curved arrows show the flow of electron or electrons during bond breaking and/or bond making…
Q: Which best describes the resonance hybrid for the following molecule? N The electron deficiency is…
A: Resonance structure: If the Lewis structure of a molecule or ion cannot explain by a single…
Q: The following table contains some data showing the effect of molar concentration of a solution on…
A: Given: The data for concentration and optical density is: Concentration (M) Optical density…
Q: Name all of the functional groups present in this structure. но. OH
A: Given compound
Q: The proton concentrations of three solutions at 25 °C are given. Classify the solutions as acidic,…
A: According to the proton concentrations given, we have to determine if the solution is acidic, basic…
Q: Nitroglycerin, a very unstable compound (molecular structure shown below), is a liquid at room…
A: Answer: In this question by using the stoichiometric correlation we have to find out the total moles…
Q: Perform a confirmational analysis on 2-bromo-3-methylbutane.
A: Given 2-bromo-3-methylbutane
Q: Read the descriptions of physical or chemical changes in the table below. Then decide whether the…
A: Thermodynamics is branch of chemistry in which we deal with amount of heat evolved or absorbed by…
Q: At 1 atm, how much energy is required to heat 55.0 g H₂O(s) at -16.0 °C to H₂O(g) at 161.0 °C? Use…
A: Answer: Converting ice from -160C to steam at 1610C is 5 step process: 1. Heating of ice 2. melting…
Q: What is the pOH of an aqueous solution of 2.56 × 10-² M sodium hydroxide? POH =
A: Given : [NaOH] = 2.56 × 10-2 M Tip : pOH = -log[OH-]
Q: You make 20.0 g of a sucrose (C₁₂H₂2O11) and NaCl mixture and dissolve it in 1.00 kg water. The…
A: According to the question, Mass of sucrose and NaCl = 20.0 g Mass of water = 1.00 kg Freezing…
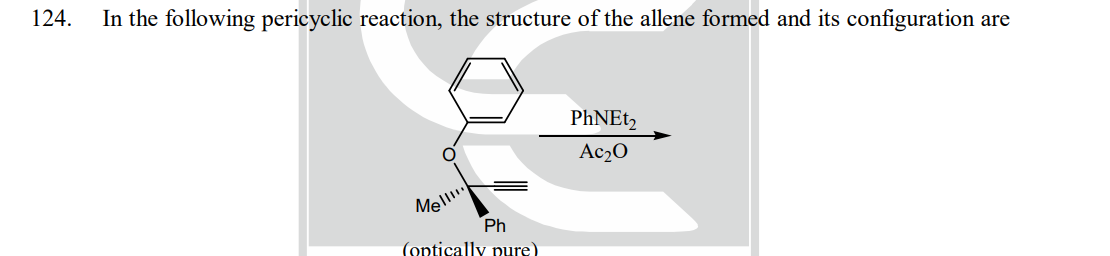
Step by step
Solved in 2 steps with 1 images

- A8 Is there anyone who can provide this pericyclic rxn mechanism?For each reaction, decide whether substitution or elimination (or both) is possible, andpredict the products you expect. Label the major products. chlorocyclohexane + NaOC(CH3)3 in (CH3)3COHN(CH2CH3)3 + HNO3 --------> a.) rewrite the reaction using bond-line structure of reagents and products of the reaction b.) supply the curved arrows explaining the mechanism of the reaction
- Elimination occurs when (Z)-3-bromohex-3-ene is treated with NaNH2. Under the same conditions, 1-bromocyclohexeneundergoes elimination much more sluggishly. Explain whyDraw all products of the reaction of (1S,2R)-1-bromo-1,2-dimethylcyclohexane in 80%H2O/20%CH3CH2OHat room temperature..Predict the major products formed when 2- methyl-1-butene reacts with: H2, Pt/25°C. Show the reaction mechanism the given alkene reactions
- There are two isomeric cyclohexa-1,4-diene products when toluene undergoes the Birch reduction (see Problem 25.24). (a) Draw the mechanism that leads to the formation of the major product. (b) Will the Birchreduction of toluene occur faster or slower than the Birch reduction of benzene itself? Hint: Is –CH3 an electron-donating or an electron-withdrawing group?Is the reagent for all elimination reactions conc. H2SO4? or just the reactions ones with OH?(CH3)3CHOH is reacted with CH3-O-C--C6H5 in a transesterification reaction. What is the M+ of the product? || O 102 80 178 None of these
- Chemistry 60. Give the principal product(s) expected, if any, when trans-1,3-pentadiene reacts under the following conditions. Assume one equivalent of each reagent reacts unless noted otherwise. (a) H2O, H3O+ (b) Na+EtO- in EtOH (c) Maleic anhydride heatPredict the stereochemistry of the following Pericyclic reactions a) a thermal [2 + 6] cycloaddition b) photochemical [3 ,6] sigmatropic rearrangementfor each reaction give the expected substitution product and predict wether the mechanism will be first order (SN1) or second order (SN2) a) 2-chloro-2-methylbutane + CH3COOH b) isobutyl bromide + NaOMe c) 1-iodo-methylcyclohexane + CH3CH2OH

