Jessica made 300 mL of primary standard using 2.35 g of sodium carbonate. She then conducted a titration to determine the concentration of her secondary standard, a sample of sulfuric acid. Her experimental results are in the table below. Trial 1 Trial 2 Trial 3 Trial 4 Trial 5 Volume of primary standard used 35.00 35.00 35.00 35.00 35.00 (ml) Initial burette 0.00 24.00 0.00 0.00 24.90 reading Final burette 24.70 48.85 24.80 24.90 47.95 reading (a) Determine the concentration of the secondary standard.
Jessica made 300 mL of primary standard using 2.35 g of sodium carbonate. She then conducted a titration to determine the concentration of her secondary standard, a sample of sulfuric acid. Her experimental results are in the table below. Trial 1 Trial 2 Trial 3 Trial 4 Trial 5 Volume of primary standard used 35.00 35.00 35.00 35.00 35.00 (ml) Initial burette 0.00 24.00 0.00 0.00 24.90 reading Final burette 24.70 48.85 24.80 24.90 47.95 reading (a) Determine the concentration of the secondary standard.
Chapter5: Errors In Chemical Analyses
Section: Chapter Questions
Problem 5.10QAP
Related questions
Question
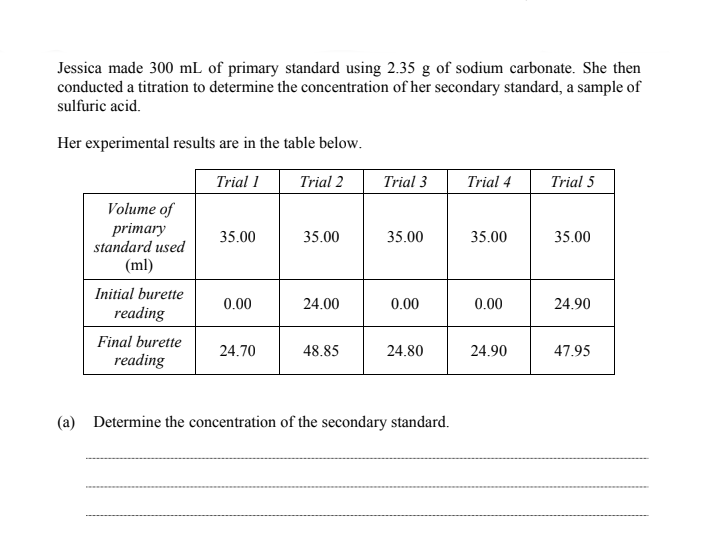
Transcribed Image Text:Jessica made 300 mL of primary standard using 2.35 g of sodium carbonate. She then
conducted a titration to determine the concentration of her secondary standard, a sample of
sulfuric acid.
Her experimental results are in the table below.
Trial 1
Trial 2
Trial 3
Trial 4
Trial 5
Volume of
primary
standard used
35.00
35.00
35.00
35.00
35.00
(ml)
Initial burette
0.00
24.00
0.00
0.00
24.90
reading
Final burette
24.70
48.85
24.80
24.90
47.95
reading
(a) Determine the concentration of the secondary standard.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

