KINETICS AND EQUILIBRIUM Dariana Using an integrated rate law for a first-order reaction At a certain temperature the rate of this reaction is first order in H CO, with a rate constant of 8 44 s H2CO3 (ag) H2O(aq)+Co, (aq) Suppose a vessel contains H,CO at a concentration of 0.990 M. Calculate the concentration of HCO3 in the vessel 0.130 seconds later. You may assume no other reaction is important. Round your answer to 2 significant digits. Ar ? X
KINETICS AND EQUILIBRIUM Dariana Using an integrated rate law for a first-order reaction At a certain temperature the rate of this reaction is first order in H CO, with a rate constant of 8 44 s H2CO3 (ag) H2O(aq)+Co, (aq) Suppose a vessel contains H,CO at a concentration of 0.990 M. Calculate the concentration of HCO3 in the vessel 0.130 seconds later. You may assume no other reaction is important. Round your answer to 2 significant digits. Ar ? X
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 5RQ
Related questions
Question
100%
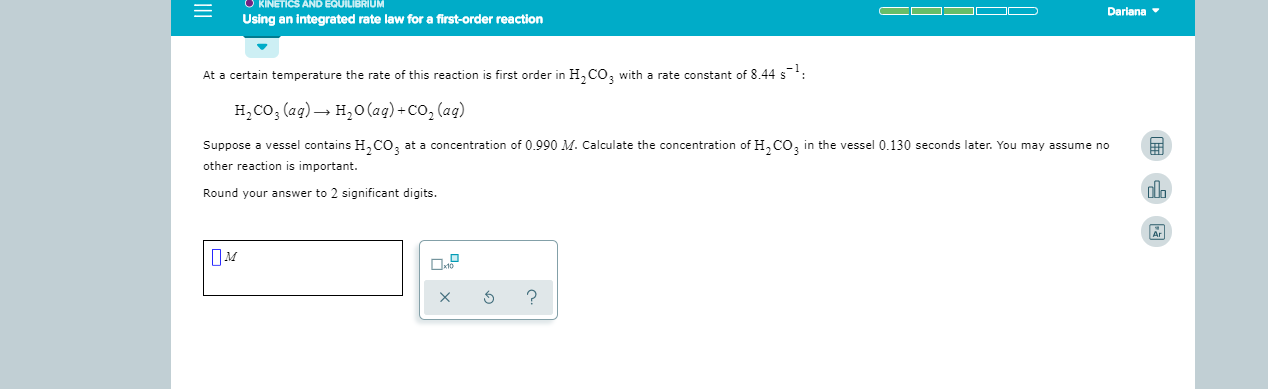
Transcribed Image Text:KINETICS AND EQUILIBRIUM
Dariana
Using an integrated rate law for a first-order reaction
At a certain temperature the rate of this reaction is first order in H CO, with a rate constant of 8 44 s
H2CO3 (ag)
H2O(aq)+Co, (aq)
Suppose a vessel contains H,CO
at a concentration of 0.990 M. Calculate the concentration of HCO3
in the vessel 0.130 seconds later. You may assume no
other reaction is important.
Round your answer to 2 significant digits.
Ar
?
X
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning