Q: Complete the balanced chemical reaction for the following weak base with a strong acid. In this case...
A: A balanced chemical equation is that equation which has all the number and types of atoms equal on t...
Q: Why do anhydrides show two carbonyl peaks?
A: The stretching frequency of any functional group can be determined by IR spectroscopy. Using IR spec...
Q: Calculate the expected ΔHrxn using the ΔHf values for the reactants andproducts in the Reaction. (Hi...
A: In an neutralisation reaction between a strong acid HCl and strong base NaOH, the acid give H+ ions...
Q: A chemist needs 3 moles of, O2 gas for a particular experiment. How many liters of O2 should she use...
A: In chemistry, when chemicals are being used for any kind of reaction, their quantity is measured and...
Q: Answer 36 only!! 35 is for reference!!
A: The location of the new bond is identified in the figure below :
Q: 4. It is suspected that a water-insoluble solid unknown is an amine. Which of the following would be...
A: Introduction: Amines are the organic compounds consist of nitrogen having a lone pair. The functiona...
Q: Plz show work thanks
A: Since you have asked multiple question ,we will solve the first question for you. To get the remaini...
Q: 00.0 mL of a 0.885 M soluti f KBr is diluted to 500.0 mL. Vhat is the new concentratic f the solutio...
A: The question is based on the concept of solutions. molarity is a method of expressing concentratio...
Q: As a genetic engineer, you have been given the following task:alter a protein’s structure by convert...
A:
Q: QUESTION 25 Dehydration WILL NOT produce the desired product from the given starting material. (See ...
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic addit...
Q: For each of the following unbalanced chemical equations,write the corresponding chemical statement.(...
A: (a) Unbalanced equation: S8 + O2 → SO2 Chemical statement: sulfur burns in presence of oxygen to fo...
Q: Painful urination is called _______________.
A: Painful urination can occur in a person of any age due to several reasons. The most common reason is...
Q: What are the oxidation numbers of each element in the arsenate ion, AsO₄³⁻? A) As = 0, O = -3 ...
A: Introduction: Oxidation number is the number of electrons that are removed or added while forming ch...
Q: Consider the following redox reaction: Ag⁺(aq) + Fe²⁺(aq) ⇌ Ag(s) + Fe³⁺(aq). How many moles of elec...
A: Fear the reaction is Redox reaction is given Redox reaction means oxidation and reduction both will ...
Q: 5.A student obtained 11.4 g of PbSO4 from the following reaction. The student added 15 g of Lead (II...
A:
Q: What masses of dinmethylamine and dimethylammonium chloride do you need to prepare 6.00 L of pH = 12...
A:
Q: iron (ii) hydroxide, Fe(OH)2 has a Ksp value equal to 4.87 x 10^-17. what is the moalr solubility of...
A:
Q: H2C2O4.2H2O + K2Cr2O7 → K[Cr(C2O4)2(H2O)2].2H2O What is the role of oxalate ion in this reaction oth...
A: Given reaction; H2C2O4.2H2O + K2Cr2O7 → K[Cr(C2O4)2(H2O)2].2H2O On balancing the equation; 7H2C2O4.2...
Q: Carbons 1 and 4 of 1,3-cyclopentadiene are equivalent and give the same carbocation on protonation. ...
A: The structure of 1,3-cyclopentadiene is as follows:
Q: 3. An unknown salt gives a red solution when reacted with KSCN, and a pale yellow ppt. when treated ...
A: Given: Iron salt when react with KSCN gives red solution. and a pale yellow ppt. when treated with A...
Q: How can the Molarity be calculated?
A: Molarity can be defined as the moles of solute per litre of the solution. Its unit is mol/L. It is d...
Q: Answer number 1.
A: HCP unit cell has 6 atoms per unit cell. And density is defined as- Density = massVolume occupied
Q: For the following reaction, 65.1g of barium hydroxide are allowed to react with 40.9g of sulfuric ac...
A: To solve this problem we have to use the Stoichiometry of balanced chemical equation. So, First we ...
Q: ewavelength of a light that emits 4.85 x 10-19 joules of energy is nm e color of that light is:
A: The question is based on the concept of atomic structure. we have to calculate the wavelength of the...
Q: Give the IUPAC name for cach of the following CH,CH;CH;CH2-COCH;CH JUPAC name C-0-CHCH, CH, CH IUPAC...
A: The International Union of Pure and Applied Chemistry, IUPAC is an authority that has set certain ru...
Q: Use arrows (up / down) to indicate the affect of each of these disturbances (stresses) on the concen...
A: Using Le Chatelier's Principle, The reaction at equilibrium shifts towards that direction in which t...
Q: A gas effuses 1.21 times faster than propane (C3H8) at the same temperature and pressure. What is th...
A:
Q: Please help solve these problems.
A: The question is based on the concept of equilibrium. we have to answer the following questions based...
Q: What is the wavelength of the photons emitted by hydrogen atoms when they undergo n =5 to n =4 trans...
A: using the given transition n=4 to n=5 apply the formula and calculate the wavelength R=109677.6cm-...
Q: Identify the organic functional groups and reaction type for the following reaction.The reactant is ...
A: We have to predict the reactant, products and reaction type
Q: An electric current transports 76.0 C of charge in 44.0 minutes. Calculate the size of the electric...
A: The size of electric current is decided by the charge passed per second.
Q: Define physical property and chemical property. Identify each type of property in the following stat...
A: Any object with a known chemical structure is referred to as a chemical substance. Water, for exampl...
Q: For each system listed in the first column of the table below, decide (if possible) whether the chan...
A: Entropy is simply degree of randomness.
Q: Problem is attached! Need help! Please and thank you!
A: Acc To Dalton law of pressurePTotal = PH2O + PO2 + PN2PTotal = (0.934 + 0.934 + 0.934 ) atmPTot...
Q: What is the pH of a 0.375 L solution made with and 25.00 mL of 5.50 M HBRO (K, = 2.3 x 10 ) 2.775 g ...
A:
Q: What is a carboxylic acid? Give an example.
A: To define: carboxylic acid? give an example of carboxylic acid?
Q: Chemistry Question
A: The details solution for this reaction is provided below in attach image.
Q: Draw the organic reactant(s) for the following reaction.
A:
Q: A 45.0-mL sample of 0.66 M HA (Ka = 0.00084) is titrated with 0.67 M NAOH. What is the pH of %3D the...
A:
Q: (4,0,1,-1/2) is a valid set of quantum numbers. TRUE OR FALSE
A: For the statement provided set of quantum number is (4,0,1,-1/2) Where Principal quantum number (n...
Q: UESTION 32 Consider the carbonyl compounds shown below. Which pair of molecules will result in a Cla...
A: Applying concept of claisen Schmitt reaction.
Q: trans-Isomers have lower solubility than cis-isomers, why?
A:
Q: A sample of gas is collected in a balloon that can easily expand and contract. It should be noted th...
A: An ideal gas is one that obeys ideal gas equation at all temperatures and pressures. A perfect ideal...
Q: Choose the missing reactant(s) for the following reaction. Select all that apply.SELECT ALL THAT AP...
A:
Q: 9. Order the following solutions according to: a) its increasing acidity: A. pH x 2.00 B. [H3O + ...
A: Higher the pH, lower the acidity and higher the basicity.
Q: PET scans require fluorine-18, which is produced in a cyclotron and decays with a half-life of 1.83 ...
A: Given: Half life, T1/2 = 1.83 hrVelocity of transport, v= 60.0 miles/hrfinal Concentration of F-18 =...
Q: Explain what happens to disaccharides and polysaccharides during digestion?
A: DISACCHARIDES They are those carbohydrates that on hydrolysis give two monosaccharides units. Glycos...
Q: 5. Which of the following is a desirable characteristic of both diagnostic and therapeutic radioisot...
A: Radio isotopes has number of applications in diagnose disease and also as an effective treatment too...
Q: The radius of a xenon atom is 1.3 * 10-8cm. A 100-mL flask isfilled with Xe at a pressure of 1.0 atm...
A: Given: radius of a xenon atom is 1.3 x 10-8 cm V = 100 mL = 0.100 L pressure of 1.0 atm temperatur...
Q: Please follow directions
A:
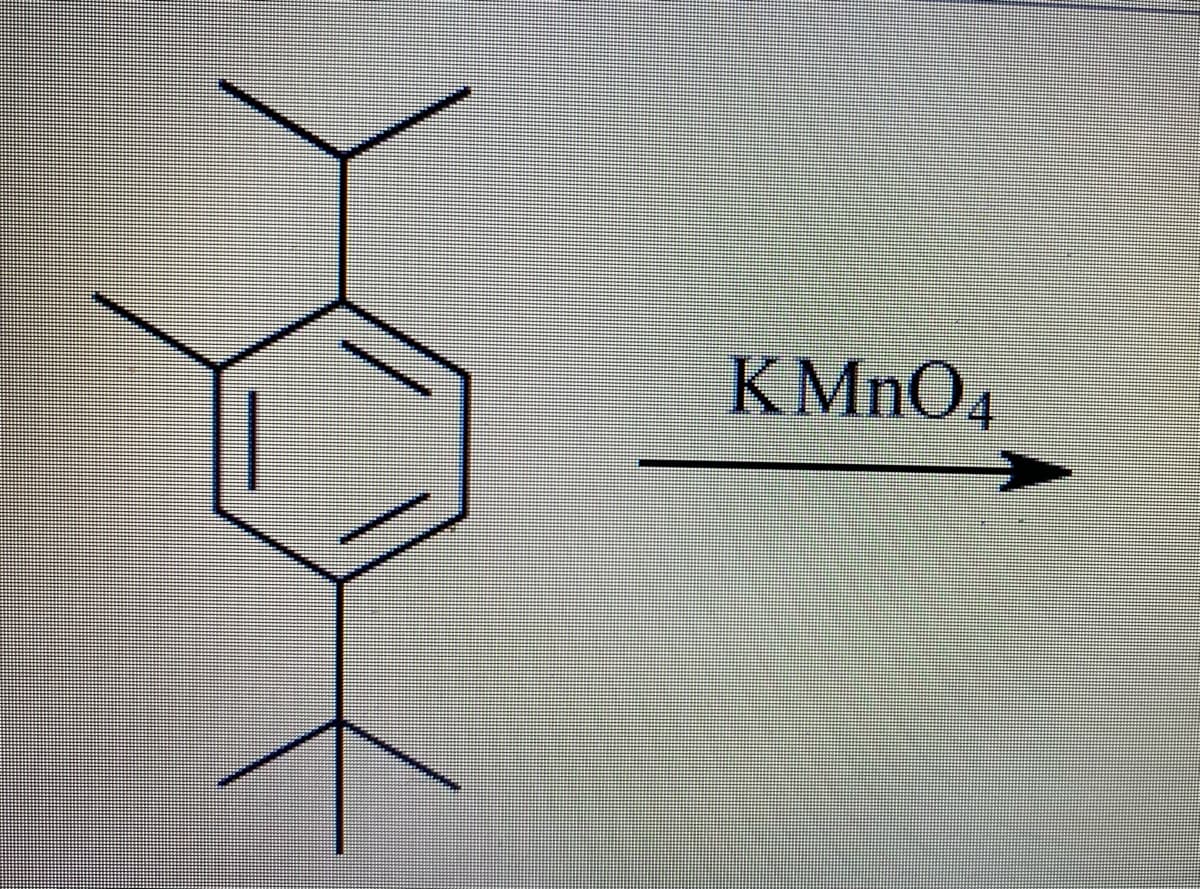
Step by step
Solved in 2 steps with 2 images


