LIMITING REAGENTS T02/S01 Based on the balanced equation Molar Mass (g/mol) 2Ba + 02 - 2Bao Ba 137,33 calculate the number of Bao formula units formed when 42 Ba atoms and 13 0, molecules react? O2 32.000 Bao 153.33 Avogadro's No. 6.022x1023 mol*1 exact number, no tolerance Question Attempts: 0 of 1 used SAVE FOR LATER SUBMIT ANSWER
LIMITING REAGENTS T02/S01 Based on the balanced equation Molar Mass (g/mol) 2Ba + 02 - 2Bao Ba 137,33 calculate the number of Bao formula units formed when 42 Ba atoms and 13 0, molecules react? O2 32.000 Bao 153.33 Avogadro's No. 6.022x1023 mol*1 exact number, no tolerance Question Attempts: 0 of 1 used SAVE FOR LATER SUBMIT ANSWER
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter3: Mass Relations In Chemistry; Stoichiometry
Section: Chapter Questions
Problem 54QAP: Ammonia reacts with a limited amount of oxygen according to the equation...
Related questions
Question
Please help
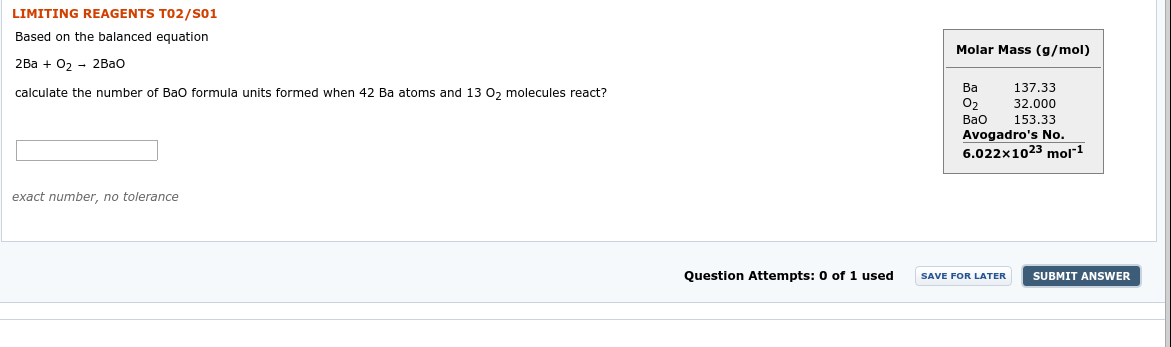
Transcribed Image Text:LIMITING REAGENTS T02/S01
Based on the balanced equation
Molar Mass (g/mol)
2Ba + 02 - 2Bao
Ba
137,33
calculate the number of Bao formula units formed when 42 Ba atoms and 13 0, molecules react?
O2
32.000
Bao
153.33
Avogadro's No.
6.022x1023 mol*1
exact number, no tolerance
Question Attempts: 0 of 1 used
SAVE FOR LATER
SUBMIT ANSWER
Expert Solution
Step 1
A mole of a substance can be defined as the mass of that substance that contains the same number of fundamental units as atoms in 12.0 g of 12C.
These fundamental units can be atoms, molecules, or formula units that depends on the substance. 1 mol of any atom, molecule or formula unit contains 6.02 x 1023 atoms, molecules or formula units.
Step 2
Given:
Number of Ba atoms = 42
Number of O2 molecules = 13 molecules
Calculate moles of Ba and O2:
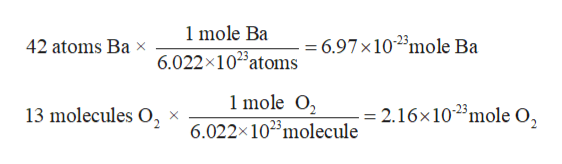
Step 3
According to balance chemical equation:
2 mole Ba = 2 moles BaO
1 mole O2 = 2 moles BaO
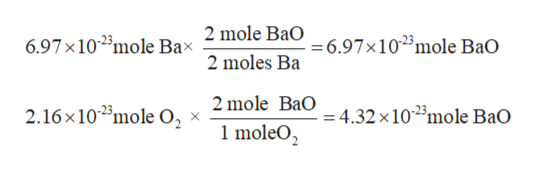
Step by step
Solved in 6 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning