m.ecollege.oom/course.htmicourselde 154322748HepiD=2b3e43e6520 d55915384a5aZ76#10001 Search. Sh. TripAdvisor (MC 13 on Temperature, Changes in Energy, and Gases Item 17 17 of 40 <> Solve for the new pressure in each of the following, with n and V constant: Review | Constants | Periodic Table Submit Request Answer Part B A sample of N, with a pressure of 780 mmHg at -70 °Cis heated to 21 °C. Express your answer using two significant figures. Ηνη ΑΣφ P= mmHg Submit Request Answer Provide Feedback Next > P Pcarson Would you like to store your password ct Us pearsoned.com? More info Yes Not for this site hp 6:55 AM 12/4/2019 ip ins prt sc f10 f6 dele 144 & bac 8. 7. T. R. Y. L. H. V. %24
m.ecollege.oom/course.htmicourselde 154322748HepiD=2b3e43e6520 d55915384a5aZ76#10001 Search. Sh. TripAdvisor (MC 13 on Temperature, Changes in Energy, and Gases Item 17 17 of 40 <> Solve for the new pressure in each of the following, with n and V constant: Review | Constants | Periodic Table Submit Request Answer Part B A sample of N, with a pressure of 780 mmHg at -70 °Cis heated to 21 °C. Express your answer using two significant figures. Ηνη ΑΣφ P= mmHg Submit Request Answer Provide Feedback Next > P Pcarson Would you like to store your password ct Us pearsoned.com? More info Yes Not for this site hp 6:55 AM 12/4/2019 ip ins prt sc f10 f6 dele 144 & bac 8. 7. T. R. Y. L. H. V. %24
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter1: Gases And The Zeroth Law Of Thermodynamics
Section: Chapter Questions
Problem 1.32E
Related questions
Question
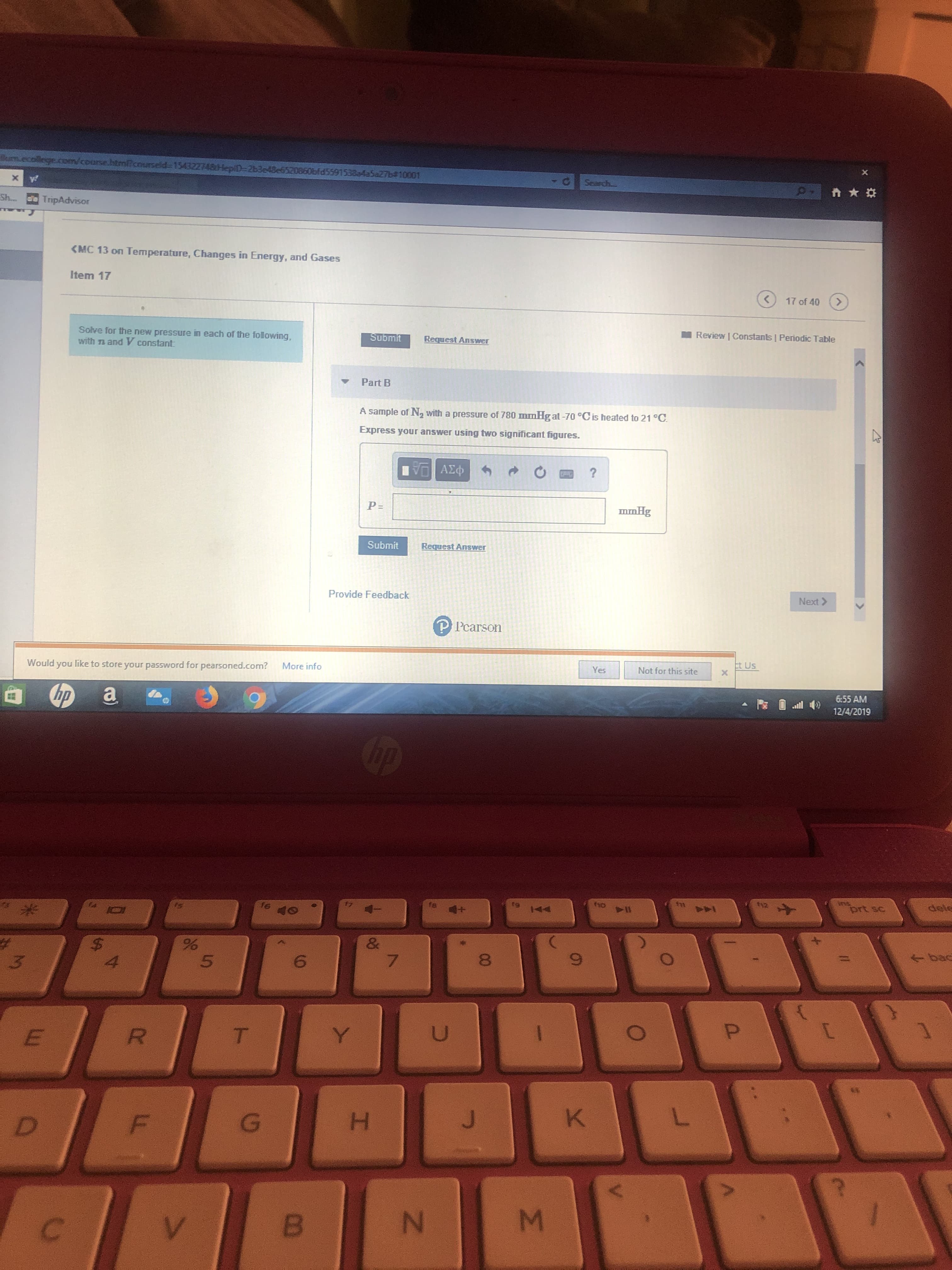
Transcribed Image Text:m.ecollege.oom/course.htmicourselde 154322748HepiD=2b3e43e6520
d55915384a5aZ76#10001
Search.
Sh.
TripAdvisor
(MC 13 on Temperature, Changes in Energy, and Gases
Item 17
17 of 40
<>
Solve for the new pressure in each of the following,
with n and V constant:
Review | Constants | Periodic Table
Submit
Request Answer
Part B
A sample of N, with a pressure of 780 mmHg at -70 °Cis heated to 21 °C.
Express your answer using two significant figures.
Ηνη ΑΣφ
P=
mmHg
Submit
Request Answer
Provide Feedback
Next >
P Pcarson
Would you like to store your password
ct Us
pearsoned.com?
More info
Yes
Not for this site
hp
6:55 AM
12/4/2019
ip
ins
prt sc
f10
f6
dele
144
&
bac
8.
7.
T.
R.
Y.
L.
H.
V.
%24
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning