Q: NH Oit 1d 25 1d 1s 25 4t 10 8. 7 6 5 1. 2. 3. 1.
A:
Q: 13.95589 183015g 17.64589 T6.99497 2.
A: Answer:
Q: A student wanted to measure the density, in g/mL, of an unknown metal alloy. They had multiple sized…
A: To find the density of unknown metal, first draw the graph of mass Vs volume. Create a regression…
Q: x2 4. (0.432)x(0.928)3 1.45 x 10-5 X-?
A:
Q: C. Pb(NO3)2(aq) + 2L¡I(aq) Pbl2 (s) + 2LİN03(aq)
A: 1- In this question, you have molecular equation and we want to write net ionic equation. You can…
Q: 8)200cm3→ dm3 9)3.5 x 105 ng → mg 0)8.43 x 1012 mg Gg
A:
Q: (e) (83.145 m - 83.142 m)/(4.52 sx 2.208 s) = i Units m/s^2 Hint Assis
A: Calculation can be done by use of addition/substraction, multiplication or division to solve for…
Q: 4 5.00 5.00 4.00 6.00 6. 3.00 7.00
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub- parts for…
Q: a. 8.7 g + 15.43 g + 19 g = 43.13 g b. 4.32 cm X 1.7 cm = 7.344 cm² c. 853.2 L - 627.443 L = 225.757…
A:
Q: 2.004, p. 3.746 + log q. 4.876 + log (,0E) = 4.632 X = '1.005 '2.005
A: The solution is given below -
Q: 35 34.9689 75.77 Chlorine 37 36.9659 24.23
A: Atomic mass is the mass of an atom. Atomic mass is sum of protons and neutrons.
Q: 4 447.9 780.7 115.5 71700 703 669 5 cm3 10 cm3 20 cm³ 09.1 726.0 411.1 796 822 20 cm3 10 cm3 5 cm3…
A: Boyle's law confirm this experiment . At constant temperature the pressure of gas is inversely…
Q: 2. Which one of the following is the greatest mass? (Hint: converting all masses to the same unit…
A: Mass is both a property of a physical body and a measure of its resistance to acceleration (a change…
Q: Perform the calculation, and report the answer to the correct number of significant figures.
A: Perform the calculation, and report the answer to the correct number of significant figures.
Q: 2NI3(s) N2(g) + 31½(g) classify:
A: We can classify reactions from the reactants and products. Chemical reactions can be of different…
Q: 10 20 30 40 50 60 70 80 90 100 g 100 200 300 400 500 g 1 2 3 6. 7 10 (A) 521.20 g (B 521.2 g © 25.20…
A: In the given picture 3 scales are there. The first two scales are mentioned as g unit. To calculate…
Q: 3. 4. 1 89 10 11 1A 2A 12 13 14 15 16 17 18 3A 4A SA 6A TA SA 1.008 He 4 4.003 LI Be 6.941 9.012 9.…
A:
Q: What is the mass of 19.8 cm3of gasoline (density 0.680 g/cm3)?
A: Given : volume of gasoline = 19.8 cm3 And density of gasoline = 0.680 g/cm3
Q: Identify each of the following atoms or ions:
A: An element can be represented as, XZA where A is the mass number and Z is the atomic number.
Q: (1) [H*] %3D 1.59 x 10-7 м (II) [H+] = 1.04 x 10-8 M 4.0 (а) pHI = 49 %3D PHII 4.0 %3D (I) [H*] 5.69…
A: pH is defined as the negative of the logarithm of the H+ concentration. pH=-logH+ pH<7 acidic…
Q: 12 32 33 18 36 39 13 11 OH OH 19 14 6 34 37 H 41 15 35 38 1021 16 17 30 22 25 26 29 28 23 24 27 F 31…
A: If the molecule contains a carbon atom attached with four different types of atoms or groups known…
Q: 3H (s) 1H (m) 2H (d) 2H (d) 1H (s) 8 7 6 5 4 3 2 1
A:
Q: 1) NaH HO. 2) A) I OH B) II C) II D) IV II IV 50% 34°F a here to search
A:
Q: ꜱᴜᴘᴘᴏꜱᴇ ᴛʜᴀᴛ ᴛʜᴇʀᴇ ᴀʀᴇ ꜰᴏᴜʀ ᴄᴏʟᴏʀᴇᴅ ᴄᴀɴᴅɪᴇꜱ ɪɴ ᴀ ʙᴏx, ᴛᴡᴏ ʀᴇᴅ ᴀɴᴅ ᴛᴡᴏ ʙʟᴜᴇ, ᴡʜᴀᴛ ɪꜱ ᴛʜᴇ ᴘʀᴏʙᴀʙɪʟɪᴛʏ…
A: Answer 4 :- Total number of Candies in the box = 4 Total number of red candies in the box = 2 Total…
Q: [(2.82 × 10®) ÷ 50.882] + 154.08
A:
Q: What is 1.3 x 0.8
A:
Q: 1. Convert the following Temperature to fill in the blanks. K °C °F а. 22.19 b. -30.55 с. 547.8
A: For the conversion of temperature, we use the following equations:
Q: 184 W 74 110 149 100 90 80 70 60 = 1.28 50 p. 40 56 26 Fe 30 1.15 20 10 78 Ne 20 10 %3D 40 20
A: A plot of the number of neutrons verses the number of protons for stable nuclei reveals that the…
Q: 5. (44.8±.7)x (98.66 ±.05)%3=
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: a) e= 4 てキこアル Mg=-12 %3D ヘ=z Ma=+3 Mg= t/z C) e =3 Me=-3 Mg n ナ/2 ms D) e=3 Me=-3
A:
Q: 2. Homogeneous mixtures(s) A. distilled water B. gasoline C. sandy water D. copper turnings + iron…
A: A. distilled water is a pure substance, not a homogeneous mixture. B. gasoline is homogeneous…
Q: HO HO Compound A Compound B Compound C Compound D 100 2112 80 1644 60 2934 %T 40 3001 2987 3397 20…
A: Given information is,
Q: The mass of a competition frisbee is 125 g. Convert its mass to ounces using the unit conversion…
A: Mass can be expressed in different units which are ounces, grams or pounds. One ounce is equal to…
Q: What is the mass of a rectangular piece of aluminum 160.6 cm * 140.8 cm * 15.2 cm The density of…
A: Volume (V) = 160.6 cm × 140.8 cm × 15.2 cm = 343709.696 cm3 Density (d) = 2.70 g/cm3
Q: 1) ТСРВА 2) а. „MgBr H,C CH2 H;C CH3 b. H30* 3) Н2, Раc H;C CH3
A: Hydrogen in presence of Pd and C behaves as a reducing agent. mCPBA is a peroxy-carboxylic acid used…
Q: b) the mass of used acetylene in pounds 0.734 0.434 0.299
A: 40 inches long and 6 inches diameter a drum contains acetylene at a initial pressure of 250 psia…
Q: O 19.0 cm³ 6.0 cm³ O 7.0 cm³ O 13.0 cm³ O
A:
Q: I - Xe 135 135 54 Xe + а. 53 b. K 40 iß + X 19 I + in 5 Mn + X 135 C. 53 25 I + on 135, 524 235 99…
A:
Q: c) A metal sample has a mass of 7.56 g. The sample is placed into a graduated cylinder previously…
A: Given : Mass of metal=7.56 g Initial volume=20.00 mL The final volume of water= 21.68 mL Volume of…
Q: V Kc 7.30 0.959 0.650 8.16 1.062 3.49 11.33 0.688 3.35 1.734 0.700 3.24
A: A) CHCl2COOC5H11 ⇔ CHCl2COOH + C5H10 Initial moles…
Q: 78.084 mL X 20. mL = g 868.04 g 72.01 mL %3D mL mol 539.4 mol 42.6 L = L
A:
Q: 1. 6C 7 N+ 14 1N 1H 0. le 0. -le
A: In a successful nuclear reaction , mass number and atomic number will be same on both reactant as…
Q: 100.0 95 3426.4 1927 66 61728 90. 479.97 243.99 1644.3 1493. 36.76 85 IS2 54 299935 80 822.53 965 45…
A:
Q: 14.98+27,340+87.7593
A: Firstly, dd the small values i.e 87.7593 and 14.8, followed by adding 27,340
Q: How do i fill in this table?
A: Density = mass (g)/volume (ml)density of water = 0.997770 g/mlHere, we have mass of material…
Q: 483.7K equals 210.7°F 756.7°F O 756.7°C 870.7°C 106
A: The question is related to conversion of temperature in centigrade in to temperature in Kelvin,…
Q: A student combines the solutions below: Calculate the [H3O+] and [OH−] in the resulting solution.
A: Given solutions are: 0.80M HCl 1.2M NaOH 0.60 M HI. What are the [H3O+] and [OH-] in the resulting…
Q: 13 12 11 10 9 8 7 6 5 4 3 2 1 0 メ。 HO, 13 12 11 10 9 8 7 6 5 4 3 1
A:
Q: 1= = exactly 36 in) = in nL = cm L(1qt=946 mL) kg = OZ exactly 1 lb and 1kg=2.20 lb) _qt (1qt=…
A:
Q: QUESTION 14 5.621 lb = grams. (1 lb = 454 g) %3D O a. 1.70 x 10 O b. 1.28 x 105 Oc 1.24 x 10-2 O d.…
A: Mass of any compound can be changed to different unit .
What are the retention times and areas of all the attached peaks?
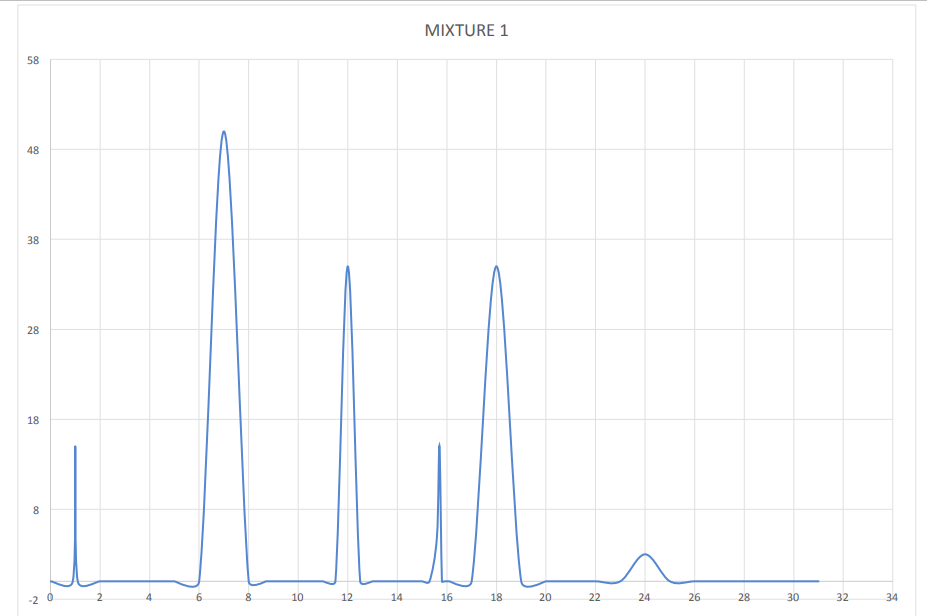
Step by step
Solved in 2 steps

- V26Which of the following values for R2 shows the strongest correlation between the X and Y values in a data set? 0.0184 0.0047 0.500 0.981 0.9678₃₀. ɪꜰ ʏᴏᴜʀ ʙɪʀᴛʜᴅᴀʏ ɪꜱ ᴏɴ ᴀ ᴍᴏɴᴅᴀʏ ᴛʜɪꜱ ʏᴇᴀʀ ᴀɴᴅ ɴᴇɪᴛʜᴇʀ ᴛʜɪꜱ ʏᴇᴀʀ ɴᴏʀ ᴛʜᴇ ɴᴇxᴛ ʏᴇᴀʀ ɪꜱ ᴀ ʟᴇᴀᴘʏᴇᴀʀ, ᴡʜᴀᴛ ᴅᴀʏ ᴏꜰ ᴛʜᴇ ᴡᴇᴇᴋ ᴡɪʟʟ ʏᴏᴜʀ ʙɪʀᴛʜᴅᴀʏ ʙᴇ ᴏɴ ɴᴇxᴛ ʏᴇᴀʀ?
- How do you convert 18.8 CP2/TM3 to YY2/ZW3? Use the folowing inequalities: 1CP=2.104 BX, *Bx=4 YY, 1TM=7.1088DD, 1DD=4.966ZW. When writing out the answer leave out the units.What volume of 95.0% alcohol by weight (density: 0.809g/cm^3) must be used to prepare150 cm^3 of 30.0% alcohol by weight (density: 0.957g/cm^3)2 ᴀᴄᴛɪᴠɪᴛʏ ₃: ʀᴇᴀᴄᴛɪᴏɴꜱ ᴏʀ ᴛᴇᴄʜɴɪQᴜᴇꜱ ɪɴ ʏɪᴇʟᴅɪɴɢ ᴏᴄ!ᴅɪʀᴇᴄᴛɪᴏɴꜱ: ɪᴅᴇɴᴛɪꜰʏ ᴛʜᴇ ʀᴇᴀᴄᴛɪᴏɴ ᴏʀ ᴘʀᴏᴄᴇꜱꜱ ʙᴇɪɴɢ ᴅᴇꜱᴄʀɪʙᴇᴅ. _________________ 1. ɪɴ ʟᴀʙᴏʀᴀᴛᴏʀʏ, ɪᴛ ɪꜱ ᴜꜱᴜᴀʟʟʏ ᴄᴀʀʀɪᴇᴅ ᴏᴜᴛ ᴡɪᴛʜ ᴀᴄᴇᴛʏʟ ᴀɴʜʏᴅʀɪᴅᴇ._________________ 2. ɪᴛ ɪꜱ ʀᴇᴀᴄᴛɪᴏɴ ᴛʜᴀᴛ ɪꜱ ᴜꜱᴇᴅ ᴛᴏ ᴏʙᴛᴀɪɴ ᴇꜱᴛᴇʀꜱ ʙʏ ᴛʜᴇ ʜʏᴅʀᴏxʏʟ ɢʀᴏᴜᴘᴏꜰ ᴛʜᴇ ᴄᴀʀʙᴏxʏʟɪᴄ ᴀᴄɪᴅ ᴀᴄᴛꜱ ᴀꜱ ᴀ ʟᴇᴀᴠɪɴɢ ɢʀᴏᴜᴘ ᴀɴᴅ ꜰᴏʀᴍꜱ ᴛʜᴇᴡᴀᴛᴇʀ ᴍᴏʟᴇᴄᴜʟᴇ ɪɴ ᴛʜᴇ ꜰɪɴᴀʟ ᴘʀᴏᴅᴜᴄᴛ. ɪᴛ ɪꜱ ʀᴇᴘʟᴀᴄᴇᴅ ʙʏ ᴛʜᴇ -ᴏʀɢʀᴏᴜᴘ ꜰʀᴏᴍ ᴛʜᴇ ᴀʟᴄᴏʜᴏʟ._________________ 3. ɪᴛ ɪꜱ ᴀ ᴍᴇᴛᴀʙᴏʟɪᴄ ᴘʀᴏᴄᴇꜱꜱ ɪɴ ᴡʜɪᴄʜ ᴀɴ ᴏʀɢᴀɴɪꜱᴍ ᴄᴏɴᴠᴇʀᴛꜱ ᴀᴄᴀʀʙᴏʜʏᴅʀᴀᴛᴇ, ꜱᴜᴄʜ ᴀꜱ ꜱᴛᴀʀᴄʜ ᴏʀ ᴀ ꜱᴜɢᴀʀ, ɪɴᴛᴏ ᴀɴ ᴀʟᴄᴏʜᴏʟ ᴏʀᴀɴ ᴀᴄɪᴅ. _________________ 4. ᴛʜɪꜱ ʀᴇᴀᴄᴛɪᴏɴ ɪꜱ ᴅᴏɴᴇ ᴛʜʀᴏᴜɢʜ ʀᴇᴍᴏᴠᴀʟ ᴏꜰ ᴡᴀᴛᴇʀ ᴀɴᴅ ᴍᴀʏ ʏɪᴇʟᴅ ᴀɴ ᴀʟᴋᴇɴᴇ ᴀɴᴅ ᴀ ᴍᴏʟᴇᴄᴜʟᴇ ᴏꜰ ᴡᴀᴛᴇʀ. _________________ 5. ɪᴛ ɪꜱ ᴀ ᴘʀᴏᴄᴇꜱꜱ ʙʏ ᴡʜɪᴄʜ ᴄᴏᴍᴘᴏɴᴇɴᴛꜱ ɪɴ ᴀ ᴄʜᴇᴍɪᴄᴀʟ ᴍɪxᴛᴜʀᴇ ᴀʀᴇꜱᴇᴘᴀʀᴀᴛᴇᴅ ɪɴᴛᴏ ᴅɪꜰꜰᴇʀᴇɴᴛ ᴘᴀʀᴛꜱ (ᴄᴀʟʟᴇᴅ ꜰʀᴀᴄᴛɪᴏɴꜱ) ᴀᴄᴄᴏʀᴅɪɴɢ ᴛᴏᴛʜᴇɪʀ ᴅɪꜰꜰᴇʀᴇɴᴛ ʙᴏɪʟɪɴɢ ᴘᴏɪɴᴛꜱ.
- What is the mean (average) value for the following data: 25.12, 25.29, 24.95, and 25.55?Convert 50.08F to:a. 8C b. KA. B. and C. already solved (https://www.bartleby.com/questions-and-answers/chemistry-question/a89691d6-2162-4677-b0cc-dc84fcceda08). Please answer E, F and GCircle in pencil in the image on th eirght cide of the diagram is a "2"( Pb(NO3)2 ).

