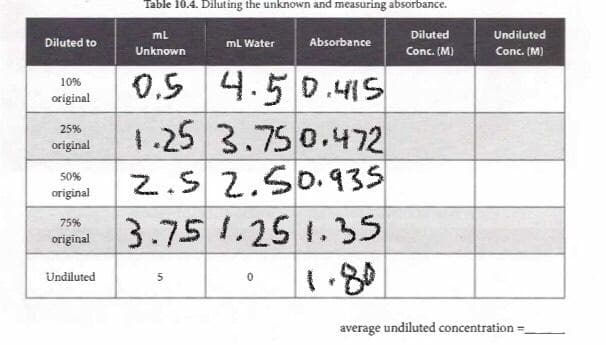
ml Diluted Undiluted Diluted to mL Water Absorbance Unknown Conc. (M) Conc. (M) 0.5 4.5 0.4IS 1.25 3.750.472 2.S 2.50.935 10% original 25% original 50% original 3.75 1.25 1.35 75% original 1.80 Undiluted average undiluted concentration =
Q: How many grams of NaAsO, should be weighed out to prepare a stock solution of 100 mg/L in 500 mL…
A: A numerical problem based on stock preparation, which is to be accomplished.
Q: 3. Calculate the mass of AgCl that can be dissolved in 10 mL of water. The Ksp for Silver Chloride…
A: We are having the Ksp as 1.83*10-10 , volume of water as 10 ml, salt is AgCl. We have to calculate…
Q: Question 8 What is the correct burette reading? 15.5 mL 15.42 mL 16.57 mL 16.6 mL
A: To measure the reading correctly we should observed the burette at eye level straight to the bottom…
Q: Dosage Form Active Raw Materials Actual Yield Formulation Packaging Ingredient 2.6 kgs paracetamol…
A: In this question, balanced chemical equation of each item is to be written.
Q: Table 8.7 (data) HCI plug location 10 NH3 plug location 60 Deposit location 32…
A:
Q: An anthocyanin sample has a concentration of 3.2874 x 10 M. Jane does a 1/300 dilution of this…
A: Dilution is the process of mixing the sample with a large volume of sterile water or other…
Q: Preparing and Diluting Solutions Data Table Test Tube 1* 2 4 10ML/3.8mL 2.4mL SmL 1.53ML…
A: We are given the data for the concentration of different diluted stock solutions and their…
Q: els Part IV: Iron content ofan unknown well water sample 34 lowo Unknown Well Water Code Unknown…
A:
Q: PART ONE: Preparation of FECNS Solution Prepare the two solutions in table 1 by accurately measuring…
A: In part 1, when 1 ml of 0.0020 M CNS- solution is added to the unused portion of solution 1 in the…
Q: . Mixing a solution of higher concentration with an identical solution of lesser concentration 2.…
A: We are authorised to solve only one question at a time. Please post rest of the questions…
Q: Test of the Purity of the Synthesized Aspirin Initial mass of aliquot of product (g) 0.210g…
A: The purity percentage is the amount of pure substance present in the total impure sample. It is…
Q: PROCEDURES: A) Preparation of three Standard ASA solutions. 1) These solutions will be prepared…
A:
Q: 3What
A: In order to determine the ratio strength, let us first calculate the amount of benzalkonium chloride…
Q: olo Data PbCl2 1.70x10-5 PBCO3 7.40x10-14 PbCrO4 2.00x10-14 PBF2 3.3x10-8 PbI2 9.8×10-9 Pb(IO3)2…
A:
Q: Table 3.4. Standardization of NaOH solution. Conditions Trial 1 Trial 2 Primary standard Weight of…
A: The neutralization of KHP (potassium hydrogen phthalate) using NaOH occurs when the gram equivalents…
Q: Seoyeon needs to prepare a 250 mL solution of 0.1000 M Na2CO3. Which glassware should she use to…
A: All the instruments mentioned can be used to measure the volume of the solution. Although for…
Q: Part B: Conductivity of pure water, solids and solutions. Substance Conductivity Substance…
A: A question based on conductivity of materials, which is to be accomplished.
Q: Which of the ff. is equivalent to the overall concentration of Mg-EDTA near the endpoint? [Mg2*]…
A:
Q: Preparation and Standardization of KMNO4 solution Experimental data Complete the table below. Trial…
A:
Q: Is this calculation correct? See image below....
A: The calculation is correct.
Q: If you have 155 mL solution of a 0.762 M FeCb solution, how many g FeCb are contained in this…
A:
Q: 3.0 Spectrum 1 2.8 2.6 2.4 2.2 2.07 1.00 B 2.0 1.8 1.6 1.4 1.19 1.2 a A 1.0 0.8 0.6 0.4 0.2 ppm
A: INTRODUCTION: 1-H NMR spectroscopy can be determined by the help of chemical shift value and the…
Q: Your NaOH concentration 0.10 Trial 1 Trial 2 Volume of Acid (in mL) 12 ml 14 ml Initial Buret…
A: Disclaimer : To find out the percentage error , actual concentration of the acid should be known.…
Q: Draw a bar diagram on your sketch paper based on the table below and determine the non-carbon…
A: Hardness is defined as the soap consuming capacity of water. Temporary hardness is caused due to…
Q: A lab pair’s Beer’s Law calibration graph 0.08 < A < 0.79 had a best-fit line of A = 0.013 C –…
A:
Q: Active Dosage Form Raw Materials Actual Yield Formulation Packaging Ingredient 500 mg per 3 kgs.…
A: In a balanced chemical reaction number of all the atoms of various elements present in the reaction…
Q: A. Standardization of NaOH Solution TRIAL [HCI] Volume Moles Moles Volume [NAOH] HCI НОEN 25.75 IOH…
A:
Q: Stock or starting Serial dilutions done Total dilution factor Final concentration concentration…
A: For first we are provided starting concentration and total dilution factor. We are required to find…
Q: Objectives: To determine the percent mass of acetic acid in vinegar Data: Vinegar Sample : Trial 1…
A: We are given the experimental data and we have to calculate the mass % of acetic acid in vinegar…
Q: AP Question // Q 5) Using the graph below, find the slope (and its unit). What letter(s) the Beer's…
A: In order to find the slope, we have to draw a linear regression plot in an excel file and using the…
Q: r Ali is currently taking Mucogel suspension at a daily dose of 10 ml after her three meals and…
A: Given : Daily dose of mucogel = 10 ml after three meals and bedtime magnesium hydroxide = 195 mg…
Q: Based on the graph below, 1. molar concentration of HCO3- 2. alkalinity in ppm CaCO3
A: Step1: Here, the HCl is added to CaCO3. So, the reaction will be shown as Here, CaCO3 and HCl react…
Q: What is the correct burette reading? 25.3 mL 24.15 mL 25.85 mL 24.1 mL
A: In volumetric analysis the burette is used to measure the volume of the solution which is filled in…
Q: Table 5: Data for Trial 3 Water displaced in mL 2 mL Time (seconds) 53 4 mL 100 6 mL 139 8 mL 178 10…
A: The order of the reaction is the sum of the exponents of the concentration terms in the…
Q: Question is attached
A: Molarity is defined as the moles of solute present per liter of the solution. It is expressed in…
Q: Please give the full calculation + explanation for each step.
A: The formula for percent by mass or mass percent is given as :
Q: Average volume NaOH used (mL) Average volume N2OH used (L) Moles NaOH used (from avg volume) Moles…
A: To find the mass and percentage of acetic acid in vinegar, we used law of chemical equivalence for…
Q: CALULATE FINL CONCENTRATION USING 2 IG FIGS AND APPROPRIATE UNITS A 6.0 mLmsample of a 50.0 %…
A: Given - Volume of acetic acid solution = 6.0 mL % (m/v) of acetic acid solution = 50.0% Final…
Q: 18 g of unknown organic sample was dissolve in 758 mL of Dicloromethane (DCM). The boiling point of…
A:
Q: Run 1 Volume of Vinegar 2.00 mL Used Concentration of NaOH Initial Buret reading Final Buret reading…
A: Stoichiometry, in balanced chemical reaction is defined by the ratio at which reactants are reacting…
Q: Your NAOH concentration 0.10 Trial 1 Trial 2 Volume of Acid (in mL) 12 ml 14 ml Initial Buret…
A: Volume of NaOH required in 'Trial 1' = (24.11-6) mL = 18.11 mL Volume of NaOH required in 'Trial 2'…
Q: The aspirin we made in lab gad 125-133 melting point and I attached the IR. Base on this…
A: The structure of aspirin is: It has the following major peaks in IR spectrum: vOH = 3100-3300 cm-1…
Q: 3. Convert stock concentrations to initial concentrations and fill in Table 2: Table 2. Trial #…
A: Absorbance of a solution is proportional directly to the concentration of absorbing species provided…
Q: A 10 ml of concentrated acetic acid was added in a sufficient quantity of water to make 350 ml of…
A:
Q: Given: Mg(OH)2 (s) <--> Mg2+(aq) + 2 OH- (aq) Kc = 5.60 x 10-12 F- (aq)…
A: Given: The Kc for the following is to be determined.
Q: C) Commercial Aspirin Tablet Analysis 1) Mass of aspirin used (g) 0.088g 2) Absorbance for final…
A:
Q: Final Serum Diluted Test Tube No. Normal Initial New NSS Dilution (mL) Saline (NSS) Dilution (ID) |…
A: Concentration of the substance is a quantification of amount of solute present in a solution with…
Q: A- STANDARD PHOSPHATE SOLUTION: Standard solution 1 4 6. Conc of PO4 (ppm) O-00 O.2 0.4 0.6 0.8 1-0…
A: Calculations: Q.1: In order to find the ppm of phosphate in the unknown water sample, we have to…
Q: GRADED FORMATIVE ASSESSMENT 1. Complete the table below. Indicate the coefficient if needed.…
A:
Q: Procedure: Several graluated eylindes and a Bwette filled with different amount of water displayed.…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
V6
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- A 5.000-g soil sample was analyzed for potassium content by extracting the potassium using 10.00 mL aqueous ammonium acetate solution. Following the extraction, the soil was filtered and rinsed. The filtrate with rinsings was diluted to exactly 50.00 mL. Then, 1.00 mL of this solution was diluted to 25.00 mL, and this dilution was tested with an instrument. The concentration in this 25.00 mL was found to be3.18 ppm. What is the concentration of the potassium in the soil in ppm?What kind of samples can be analyzed by AFM? What are the applications of AFM?dipole dipole interaction between aspirin and silica explanation pls and steps also
- How can false low results due to the Hook Effect be accounted for in immunoassay testing?A supporting electrolyte is usually added to the analyte solution to minimize solution resistance.minimize solution conductivity.make the analyte less soluble in solution.increase the scan rate.os the enrgy cost to break IMFs for acetone to mix with water likely larger or smaller?
- A 18 g of unknown organic sample was dissolve in 756 mL of benzene. The boiling point of benzene was increased by 3.36oC. As the first step of analysis, determine the moecular weight of the unknow sample? Kb of benzene= 2.64oC/m Bb of benzene = 80.09 oC density of benzene = 0.874 g/mL at 25 °C Answer in whole number, no units required.1. A 25-mL specific gravity bottle was found to weigh 46.05 g when filled with water, and 52.93 g when filled with the ophthalmic solution. If the empty specific gravity bottle weighs 21.04 g, what is the specific gravity of the ophthalmic solution? a 0.784 b 0.870 c 1.149 d 1.275DATA Note: Use the videos as references except when data is provided here. Table 1. Selection of Recrystallizing Solvent. Solvents Dissolution at room temperature? (+ or −) Dissolution at elevated temperature? (+ or −) Distilled water - - Acetone + n/a 95% ethanol - + Toluene + n/a Solid sample: Naphthalene Table 2. Mass Measurements. Mass, g Sample solid 0.5021 Empty watch glass 35.7602 Watch glass with recrystallized sample 36.2485 Table 3. Melting Point Determination. Temperature, oC Appearance of Sample in Capillary Tube … Solid 78.4 Solid 79.3 Solid with signs of liquid 79.9 Solid, liquid 80.6 Liquid, solid 81.3 Liquid with signs of solid … Liquid Theoretical Melting Point, oC: 80.2°C (176.4°F) QUESTIONS Which solvent should be used as the recrystallizing solvent? Check if your chosen solvent satisfies all characteristics of a good…
- . The conductance of 0.00017 M acetic acid solution is 153 µS when applying a probe with 0.87 cm−1cell constant. The same probe measures 5.7 µS for the conductance of water. Calculate the conductivityand molar conductivity of the solution.Missing molar conductivity Given: Conductivity: 281 us x cm^-1 (microseconds/cm) Concentration of solution: 0.05555555556 M (mol/lit) Basically, molar conductivity = conductivity/concentration, but what I'm not sure of is the proper unit for the answer molar conductivity and if it will require the given conductivity to be converted to seconds and meter (or not? I'm not sure cause i've been seeing S⋅m2⋅mol−1 in the internet as the unit for molar conductivity)V 22



