Morphine is a pain medication pain of the opiate type. It acts directly on the central nervous system (CNS) to decrease the feeling of pain. Morphine is also frequently used for pain from Myocardial infarction. The molecular structure of Morphine is shown below. Please answer the following questions. HO. N-CH3 но Morphine (opium) analgesic How many Pi bonds are present in this molecule? 2. Circle 4 functional groups present in this molecule and name them. 3. Detemine the molecular formula and moleaular mass of this compound. Indicate the hybridization state of all carbon and nitrogen and oxygen atoms in this molecule. 1.
Morphine is a pain medication pain of the opiate type. It acts directly on the central nervous system (CNS) to decrease the feeling of pain. Morphine is also frequently used for pain from Myocardial infarction. The molecular structure of Morphine is shown below. Please answer the following questions. HO. N-CH3 но Morphine (opium) analgesic How many Pi bonds are present in this molecule? 2. Circle 4 functional groups present in this molecule and name them. 3. Detemine the molecular formula and moleaular mass of this compound. Indicate the hybridization state of all carbon and nitrogen and oxygen atoms in this molecule. 1.
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter9: Bonding And Molecular Structure: Orbital Hybridization And Molecular Orbitals
Section: Chapter Questions
Problem 52IL: Ethylene oxide is an intermediate in the manufacture or ethylene glycol (antifreeze) and polyester...
Related questions
Question
Can i get help on this problem please
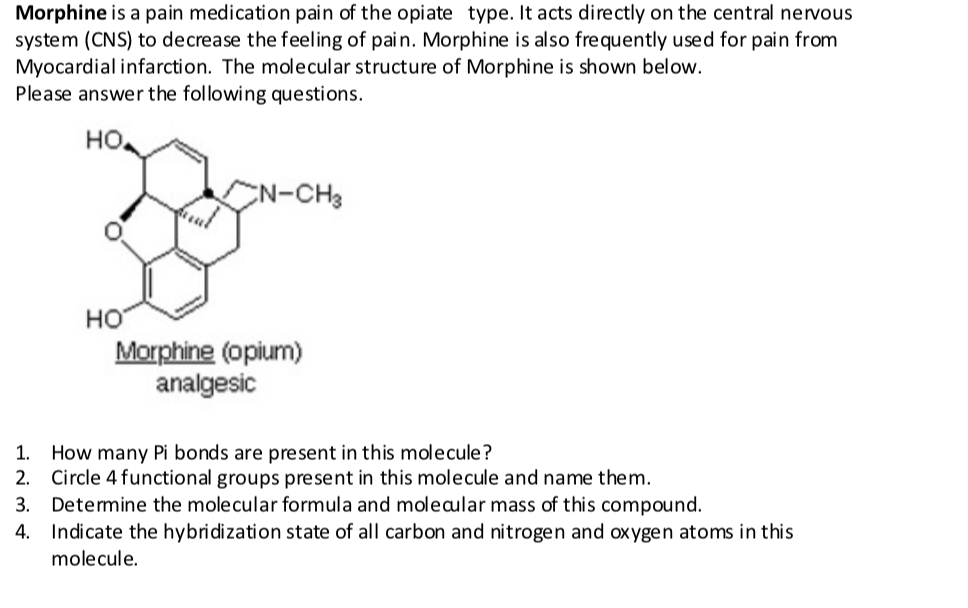
Transcribed Image Text:Morphine is a pain medication pain of the opiate type. It acts directly on the central nervous
system (CNS) to decrease the feeling of pain. Morphine is also frequently used for pain from
Myocardial infarction. The molecular structure of Morphine is shown below.
Please answer the following questions.
HO.
N-CH3
но
Morphine (opium)
analgesic
How many Pi bonds are present in this molecule?
2. Circle 4 functional groups present in this molecule and name them.
3. Detemine the molecular formula and moleaular mass of this compound.
Indicate the hybridization state of all carbon and nitrogen and oxygen atoms in this
molecule.
1.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning